Rank each of the following groups of organic compounds in order of decreasing acidity. (a) (b) (c)
Question:
Rank each of the following groups of organic compounds in order of decreasing acidity.
(a)
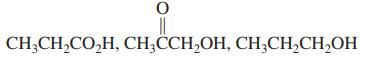
(b)
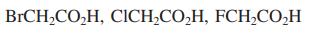
(c)
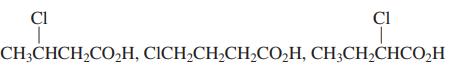
(d)
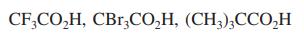
(e)
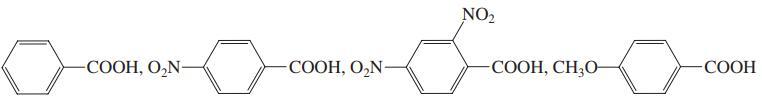
Transcribed Image Text:
CH;CH,CO,H, CH;CCH,OH, CH;CH,CH,OH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
a Pka of Propanol 16 Therefore the order decreasing acidity CH 3 CH 2 COOH CH 3 CCH 2 OH CH 3 CH 2 CH 2 OH b BrCH 2 COOH Cl CH 2 COOH FCH 2 COOH We kn...View the full answer

Answered By

SUMANT JHA
I am a Metallurgical Engineering graduate with an aggregate of 83.10%. I did my final year project from the reputed research centre of India ie, BARC on Austenitic Stainless steels.I completed my Senior Secondary Exam with an aggregate of 83.40%.
I started teaching from 2013 giving home tution to the students in Mathematics , Physics and Chemistry so till now i have seven years of experience of teaching these subjects.I also started teaching subjects of Material Science Engineering from 2018. I have assisted many students in preparing for Engineering Entrances Examination. My prime focus in tutoring is to clear the basic concepts of the subjects to the students because while i was a student i felt that clear concept is required for the deep study of the subjects.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Rank the following compounds in order of decreasing acidity of the indicated hydrogen: O 0 CH CCH2CH2CCH3 CH CCH2CH2CH2CCH3 CH CCH2CCH3
-
Rank the compounds in order of decreasing λ max: CH CH CH CH2
-
Rank each of the following sets of molecules in order of increasing SN2 reactivity. (a) CH3CH2Br, CH3Br, (CH3)2CHBr (b) (CH3)2CHCH2CH2Cl, (CH3)2CHCH2Cl, (CH3)2CHCl (c) (d) CH,CH,CI, CH,CH2I. CI...
-
A recent PwC Supply Global Chain survey indicated that companies that acknowledge the supply chain as a strategic asset achieve 70% higher performance. The Leaders in the survey point to...
-
In February 1996, President Bill Clinton signed the Telecom munications Act, which (among many other things) required all TV sets sold in the United States to be equipped with a V-chip, which allows...
-
Name some of the challenges with this traditional approach.
-
Determine whether each of the following is true or false. In each case, assume \(P\) is located at \(t=0\) and \(F\) is located at \(t=n\), and the \(A\) s are spread uniformly over the planning...
-
Sales for Hanebury Corporations just-ended year were $12 million. Sales were $6 million 5 years earlier. a. At what rate did sales grow? b. Suppose someone calculated the sales growth for Hanebury in...
-
If V f(xz, y/z), prove that zV =xVx-yVy. =
-
1. How should Captain Jones begin the process of preparing a new budget? 2. Assuming that the Greenfield officers are still relying on preventive patrol, how could a change in strategy benefit the...
-
What is the IUPAC name of the compound shown? (a) (E )-3-Methyl-2-hexenoic acid (b) (Z )-3-Methyl-2-hexenoic acid (c) (E )-3-Methyl-3-hexenoic acid (d) (Z )-3-Methyl-3-hexenoic acid H;C CO.H C=C...
-
Select the acid with the highest K a (i.e., lowest pKa). (a) H 3 CCO 2 H (b) (c) (d) (d) Cl 2 CHCO 2 H CO,H I
-
The percentage of heights between 160 cm and 188 cm Heights of Males. For Exercises, sketch the normal distribution of heights of adult males, which has a mean of 174 cm and a standard deviation of 7...
-
Ecker Company reports $1,700,000 of net income and declares $238,000 of cash dividends on its preferred stock for the year. At year- end, the company had 320,000 weighted-average shares of common...
-
Wesimann Co. issued 11-year bonds a year ago at a coupon rate of 71 percent. The bonds make semiannual payments and have a par value of $1,000. If the YTM on these bonds is 5.4 percent, what is the...
-
You, CPA, work for Anderson and Smith Chartered Professional Accountants (CPAs). It is now September 30, 2023 and you have just finished meeting with Annabelle Banks, a shareholder and the vice...
-
A train is traveling south at 71.0 m/s when the brakes are applied. It slows down with constant acceleration to a speed of 6.00 m/s in a time of 9.00 s. What is the acceleration of the train during...
-
A borrower had a loan of $ 70,000.00 at 6 % compounded annually comma with 10 annual payments. Suppose the borrower paid off the loan after 5 years. Calculate the amount needed to pay off the loan....
-
Mitchell, a calendar year taxpayer, is the sole proprietor of a fast-food restaurant. His adjusted basis for the building and the related land is $450,000. On March 4, 2014, state authorities notify...
-
SBS Company have received a contract to supply its product to a Health Care Service Hospital. The sales involve supplying 1,250 units every quarter, the sales price is RM 85 per unit. The Client...
-
Bromine is larger than chlorine, yet the two atoms have identical axial destabilization energies. Explain.
-
Draw the stereo isomers of these compounds: (a) 1, 3-Dimethyleyclohexane (b) 1, 2-Diethylcycloproane (c) 1-Chloro-3-methylcyclopentane
-
Draw both chair conformations of trans-1, 3-dimethyl cyclohexane indicate whether each methyl group is axial or equatorial.
-
4. Elizabeth has $1537.21 in her savings account. The account pays 4% compounded monthly. Elizabeth does not make any deposits or withdrawals over the next 6 months. How much interest does the...
-
7. Part of an amortization table is shown below. Determine the missing values in the table labeled A, B, C, D Payment Monthly Interest Principal Outstanding A) number payment paid paid balance 0 $175...
-
3. Calculate the following dosages according to kilogram of body weight: a. The physician ordered Zovirax capsules 5 mg/kg every 8 hours for 7 days for a patient who has a diagnosis of herpes zoster....

Study smarter with the SolutionInn App


