Reductive amination of excess formaldehyde with a primary amine leads to the formation of a dimethylated tertiary
Question:
Reductive amination of excess formaldehyde with a primary amine leads to the formation of a dimethylated tertiary amine as the product (see the following example). Propose an explanation.
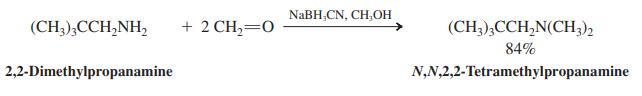
Transcribed Image Text:
NABH,CN, CH,OH (CH3),CCH,NH, + 2 CH,=0 (CH3),CCH,N(CH;), 84% 2,2-Dimethylpropanamine N,N,2,2-Tetramethylpropanamine
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (6 reviews)
The given reaction is reductive amination or reductive alkylation When 22 dimethylpro...View the full answer

Answered By

User l_359487
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Heating acetone with sulfuric acid leads to the formation of mesitylene (1, 3, 5-trimethylbenzene). Propose a mechanism for this reaction.
-
The two most general amine syntheses are the reductive amination of carbonyl compounds and the reduction of amides. Show how these techniques can be used to accomplish the following syntheses. (a)...
-
Excess ammonia must be used when a primary amine is synthesized by reductive amination. What product will be obtained if the reaction is carried out with an excess of the carbonyl compound instead?
-
A student has six textbooks, each with a thickness of 4.0 cm and a weight of 30 N. What is the minimum work the student would have to do to place all the books in a single vertical stack, starting...
-
A country has had a steady value for its floating exchange rate (stated inversely as the domestic currency price of foreign currency) for a number of years. The country now tightens up on (reduces)...
-
Explain the role education plays in hedge fund performance.
-
Dr. Walter Sullivan was one of several plastic surgeons in Las Vegas visited by Julie Jones. Jones, an exotic dancer, sought plastic surgery to improve her ability to make money in her profession....
-
Suppose the amounts presented here are basic financial information (in millions) from the 2014 annual reports of Nike and adidas. Instructions Calculate the accounts receivable turnover and average...
-
19. Derive the augmented Harrod-Domar equation: s/0 = g*+n+8. 20. What is demographic transition? Why is it relevant for the Harrod-Domar model?
-
What will your portfolio be worth in 10 years? In 20 years? When can you stop working? The Human Resources Department at Four Corners Corporation was asked to develop a financial planning model that...
-
Reductive amination of excess formaldehyde with a primary amine leads to the formation of a dimethylated tertiary amine as the product (see the following example). Propose an explanation. NABH,CN,...
-
Several of the natural amino acids are synthesized from 2-oxocarboxylic acids by an enzymecatalyzed reaction with a special coenzyme called pyridoxamine. Use electron-pushing arrows to describe each...
-
States also offer protection for consumers under the Uniform Commercial Code (UCC) and other statutes. What standard does the UCC provide for determining whether a contract is unconscionable?
-
b) Suppose instead that B-Team Inc plans to finance the investment by borrowing $150K in the third year. The firm plans to repay this debt (both interest and principal) in the fourth year. For...
-
How the Transcontinental Railroad Impacted Job Growth? it need to be a bullet point essay. It should be at least 2 pages, single spaced, 10 point Times New Roman font (the length only includes the...
-
and Define f(x) = ln(x)+In + In +In +In (3) + In (17) g(x) = ln(x)+In + In() +...+In (22020) (1) Simplify f(x), so that it is a composition of In(r) and basic transformations (shifts, stretches, and...
-
Over the last few years, the popularity of e-commerce channels such as Amazon and Flipkart has increased rapidly. How does this affect the bargaining power of logistics companies such as FedEx that...
-
The WACC K. Bell Jewelers wishes to explore the effect on its cost of capital of the rate at which the company pays taxes. The firm wishes to maintain a capital structure of 35% debt, 10% preferred...
-
Polly and her husband, Leo, file a joint return and expect to report 2014 AGI of $75,000. Polly's employer offers a child and dependent care reimbursement plan that allows up to $7,000 of qualifying...
-
Suppose Green Network Energy needs to raise money to finance its new manufacturing facility, but their CFO does not think the company is financially capable of making the periodic interest payments...
-
Show syntheses of these compounds from(Z)-2-butene: ) - a) H H C-C - CH3 . .
-
Show the products of thesereactions: . C-C CH3 1) O, CH,OH 1) O3. CH,OH a) CH;CH,CH-CH2 b) 2) (CH,),S 2) (CH,)2S CH3 . 1) 0,, CH;OH 1) 0,, CH,OH d) 2) (CH3),S 2) (CH3)2S 1) O3, CH,OH 2) (CH3),S
-
Show the alkenes that produce these compounds onozonolysis: a) CH,CCH,CH, + CH,CH b) c) CH;CCH,CH;CH + HCH + CH,CH
-
1. In an isolated calorimeter, 105 g of water is placed in a 20-g aluminum cup. Both the water and the cup start at 32 C. 15 g of ice, initially at 0 C is added to the water and the system is allowed...
-
Self-Travel, a car rental firm, has two methods of charging for car rental: Method 1 $64 per day + 25 cents per kilometre Method 2 $89 per day with unlimited travel a Write a rule for each method if...
-
18. Lets pretend that there is no air resistance. Determine the potential and kinetic energy for a 1 kg ball dropped at a height of 200 meters at for 3 seconds. SHOW ALL WORK FOR CREDIT. Time of Ball...

Study smarter with the SolutionInn App


