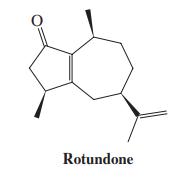
Rotundone (margin) is the natural product responsible for the peppery aroma in peppers, many herbs, and red
Question:
Rotundone (margin) is the natural product responsible for the peppery aroma in peppers, many herbs, and red wines. What cyclic diketone will give rotundone upon intramolecular aldol condensation?
Transcribed Image Text:
Rotundone
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 40% (10 reviews)
Note The molecule A is the cyclic diketone which will give rotundone upon ...View the full answer

Answered By

Vivek Chaudhary
Hi! I am vivek.My greatest passion in life is teaching. I was born and raised in india, and experienced great success at school and at university due to amazing and unforgettable teachers. This is the foundation of my commitment to helping out my students, whatever their abilities may be. Currently, I am studying a masters degree specializing in chemistry, rounding out my undergraduate background in chemistry (Honors) I have been tutoring and teaching for 2 years in various settings – tutoring small and large groups.
So, if you stuck in any problem you can ask.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Give the structure of the product derived by intramolecular aldol condensation of the keto aldehyde shown:
-
Give the structure of the product derived by intramolecular aldol condensation of the keto aldehyde shown: O CHI KOH. H-0 CH CCH CCHOCHIoO CH3
-
Give the structure of the mixed aldol condensation product of benzaldehyde with
-
a. Find the probability of getting exactly 1 girl in 10 births. b. Find the probability of getting 1 or fewer girls in 10 births. c. Which probability is relevant for determining whether 1 is an...
-
In this case, plaintiff, Carol Burnett, sued the defendant, the National Enquirer, for compensatory and punitive damages as a result of an article published in defendant magazine. The article claimed...
-
Discuss the assumptions used in modern portfolio theory (MPT) and traditional finance models.
-
Continuing to focus on evidence associated with the act, concealment, and conversion, use the evidentiary material to continue the examination. In addition, as the examiner also start to think of...
-
The Bartram-Pulley Company (BPC) must decide between two mutually exclusive investment projects. Each project costs $6,750 and has an expected life of 3 years. Annual net cash flows from each project...
-
3. Consider a two period problem where a consumer has preferences over consumption in the two periods given by: log c + Blog c'. She has no initial assets and has income y in the first period y' in...
-
Mary Gonzales is the controller at Automax, a car dealership. She recently hired Anik Cousineau as a bookkeeper. Cousineau wanted to attend a class on Excel spreadsheets, so Gonzales temporarily took...
-
Give the likely products for each of the following aldol addition reactions. NaOH, H,O () 2 -CH,CHO NaOH, H,O (b) - + (H3)CHO NaOH, H,O CH3 NaOH, H,O () H3C (d) CH3
-
Write all possible products of the base-catalyzed crossed aldol reactions between each pair of reaction partners given below. (Multiple products are possible in every case; be sure to include...
-
A 0.5-m3 vessel contains 10 kg of refrigerant-134a at -20C. Determine (a) The pressure, (b) The total internal energy, and (c) The volume occupied by the liquid phase.
-
Discuss using examples how Bamboo HR software can be used to help organizations 1. Facilitate employee's records 2. Document management 3. Employee services 4. Absence requests 5. Clocking in...
-
Should the manager or employee have the primary role in setting performance goals? Explain.
-
3. Describe a study design you could use to convince your CEO that employee attitudes are causally related to customer satisfaction scores (i.e. employee commitment leads to customer satisfaction)?...
-
Willis and Patillo discuss the "misperception" of their research subjects and the way in which misperception has reduced political awareness and effectiveness. Can you explain this conclusion? Can...
-
Provide supporting points and reference to describe the topic in terms of managing the international environment. Examine the foreign labor environment including "guest workers" and international...
-
Geoff incurred $900,000 of mining and exploration expenditures. He elects to deduct the expenditures as quickly as the tax law allows for regular income tax purposes. a. How will Geoff's treatment of...
-
Which of the following streaming TV devices does not involve use of a remote controller? A) Google Chromecast B) Apple TV C) Amazon Fire TV D) Roku
-
A gas-phase recombination reaction is first-order in each of the reactants. The energy of activation for the reaction is 49.6 kJ mol-1 at 55C the rate constant is 0.23 m' S-I. Calculate the entropy...
-
Calculate the entropy of activation for a collision between two structure less particles at 500 K, taking M = 78 g mol-3 and 0'= 0.62 nm2,
-
The pre-exponential factor for a gas-phase decomposition of ozone at low pressures is 2.3 X 1013 dm-3 mol-1 s-1 and its activation energy is 30.0 k] mol-1. What are? (a) The entropy of activation,...
-
Question 1) Billy Bob's Bikes Corporation is having some issues with their accounting department. They hired some accountants who got their educations at an institution other than Eastern Illinois...
-
A contractor or a painter who was contracted to perform a specific project. Does their service cover by the Sale of Goods act? what factors required to complete the contract in a short time. Please...
-
On average, clinic patients are expected to contribute $10 per visit, irrespective of location. Annual donations at each location are expected to be $1 million, and are expected to grow 15 percent...

Study smarter with the SolutionInn App


