Structure A (shown in the margin) is that of a naturally occurring a-amino acid. Select its name
Question:
Structure A (shown in the margin) is that of a naturally occurring a-amino acid. Select its name from the following list.
(a) Glycine;
(b) Alanine;
(c) Tyrosine;
(d) Cysteine.
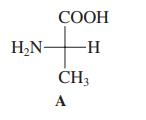
Transcribed Image Text:
СООН H,N- -H- CH3 A
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
Structure A Answer b Alanine Structure A is that of a nat...View the full answer

Answered By

Vinod Antil
I had completed my graduation (BSc Hons chemistry)from University of Delhi in2015
I am pg holder. I had completed my post graduation (MDU , Rohtak)in 2017.
I has qualified CSIR JRF NET exam and Gate exam.
From 2018 March onwards I am giving the sevice in Eureka public school ,Narnaul.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
When compound A (shown in the margin) is treated with dilute mineral acid, an isomerization takes place. Which of the following compounds is the new isomer formed? CHCH3 CHCH3 A
-
Amino acid analysis of a certain tetrapeptide gave alanine, glycine, phenylalanine, and valine in equimolar amounts. What amino acid sequences are possible for this tetrapeptide?
-
Most naturally occurring amino acids have chirality centers (the asymmetric carbon atoms) that are named (S) by the Cahn-Ingold-Prelog convention (Section 5-3). The common naturally occurring form of...
-
Troy Engines, Ltd., manufactures a variety of engines for use in heavy equipment. The company has always produced all of the necessary parts for its engines, including all of the carburetors. An...
-
In 1985, neither Florida nor Georgia had laws banning open alcohol containers in vehicle passenger compartments. By 1990, Florida had passed such a law, but Georgia had not. (i) Suppose you can...
-
Michigan Health Center, a for-profit hospital, is evaluating the purchase of new diagnostic equipment. The equipment, which costs $600,000, has an expected life of five years and an estimated pretax...
-
1. Working in small groups, complete the Pay Raise Worksheet. 2. After the class reconvenes, group spokespersons present group findings. Pay Raise Worksheet April Knepper is the new supervisor of an...
-
Amanda McNall is the corporate controller of Scott Quarry. Scott Quarry operates 12 rock-crushing plants in Scott County, Kentucky, that process huge chunks of limestone rock extracted from...
-
Question 3.1 Create a class containing a method name createArray and a main method. The method createArray creates an array where each element contains the square of its index. The size of the array...
-
Richard McCarthy (born 2/14/64; Social Security number 100-10-9090) and Christine McCarthy (born 6/1/1966; Social security number 101-21- 3434) have a 19-year-old son (born 10/2/99 Social Security...
-
Amino acids can be used as enantiomerically pure starting materials in organic synthesis. Scheme I depicts the first steps in the synthesis of a reagent employed in the preparation of...
-
The primary structure of a protein refers to: (a) Cross-links with disulfide bonds; (b) Presence of an a helix; (c) The a-amino acid sequence in the polypeptide chain; (d) The orientation of the side...
-
Calculate the length of a turn in: a. A-DNA b. B-DNA c. Z-DNA
-
The essential sections needed in an effective job description. 1. Job identification 2. Job summary 3. Responsibilities and duties 4. Authority of incumbent 5. Standards of performance 6. Working...
-
if a company has $100,000 in revenue, $20,000 in equipment depreciation and $10,000 in deductions, what is their taxable income?
-
BMW's strategic analysis of the transition to electric vehicles. What alternatives are there for consumers and for the BWM to be successful?
-
Based on Customer Services, what is flower of service and give example with Malaysia context?
-
Calculate the flow rate of blood (of density 1.02 g/cm3) in an aorta with a cross-sectional area of 1.97 cm2 if the flow speed is 40.7 cm/s .
-
Doug incurred and paid the following expenses during the year: $50 for a ticket for running a red light while he was commuting to work. $100 for a ticket for parking in a handicapped parking space....
-
A city maintains a solid waste landfill that was 12 percent filled at the end of Year 1 and 26 percent filled at the end of Year 2. During those periods, the government estimated that total closure...
-
Reaction of HBr with 2-methyipropenc yields 2-hromo-2-methylpropane, what is the structure of the carbocation formed during the reaction? Show the mechanism of the reaction. CH CHBr C=CH2 + HBr H...
-
Add curved arrows to the following polar reactions to indicate the flow of electrons in each: :Ci: (a) - :- + -N :CI: -NH (b) H CH: -Br: :Br: (c) :0: :CI: CI "
-
Predict the products of the following polar reaction, a step in the citric acid cycle for food metabolism, by interpreting the flow of electrons indicated by the curve darrows: H2 CO2 Lc-co, -0,C-CH2
-
Which of the following methods are used to configure node based backend server application and front end react application Question 4 options: Add proxy property inside react application package.json...
-
An object's momentum changed from (-5.0. 1) kg m/s to (14,-14,-4) kg m/s in 1.6s. The object's mass is 5.5 kg. How much work was done on this object during this time? work-
-
A wave with the following deep-water characteristics is propagating toward the shore in an area where the bottom contours are all straight and parallel to the coastline: Ho=3m, T= 10s The bottom is...

Study smarter with the SolutionInn App


