Typical conditions for nitrosation are illustrated in the following equation. Propose a detailed mechanism for this reaction.
Question:
Typical conditions for nitrosation are illustrated in the following equation. Propose a detailed mechanism for this reaction.
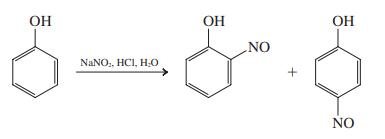
Transcribed Image Text:
OH OH ОН NO NANO,, HCI, H0 NO
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 81% (11 reviews)
Mechanism OH No nitrexsation Is th...View the full answer

Answered By

Afsar Ahmed
Hello. I am Afsar. I have been graduated in Chemistry in the year 2018. I am also a student and pursuing masters (M.Sc) & hopefully will be completing the degree in the 2020. I will be happy to solve problems in the concerned subject that I have been studying for last few years .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Propose a mechanism for each reaction. (a) (b) Ht H3C CH3 OH H SOA Ph Ph Ph Ph
-
Propose a mechanism for each reaction. (a) (b) (c) (d) OH H2SO heat OCH + CH3OH H20 CH2OH H2So4 heat CH OCH2CH CH CH,CH2OH (a minor product)
-
(a) Mitomycin is a clinically used antitumor antibiotic that acts by disrupting DNA synthesis through covalent bondforming reactions with deoxyguanosine in DNA. Maria Tomasz (Hunter College) and...
-
Craps is a dice game in which two fair dice are cast. If the roller shoots a 7 or 11 on the rst roll, he or she wins. If the roller shoots a 2, 3, or 12 on the rst roll, he or she loses. (a) Compute...
-
Wells filed a lawsuit against her employer Clackamas Gastorenterology Associates, P.C. alleging that the medical clinic violated the Americans with Disabilities Act of 1990 (ADA or Act) when it...
-
The diagram depicted in Figure 1.39 is an automatic closed-loop system for paper moisture level control. Desired Moisture Controller Drier moisture level level Moisture meter Figure 1.39 A...
-
What is the expectations gap?
-
Jamison Woodworking uses normal costing and allocates manufacturing overhead to jobs based on a budgeted labor-hour rate and actual direct labor-hours. Under-or overallocated overhead, if immaterial,...
-
- Consider an asymmetric Cournot duopoly game, where the two firms have different costs of production. Firm 1 selects quantity 91 at a pro- duction cost of 2q. Firm 2 selects quantity 92 and pays the...
-
A computer company that recently introduced a new software product claims that the mean time taken to learn how to use this software is not more than 2 hours for people who are somewhat familiar with...
-
The nitroso group, NO, as a substituent on a benzene ring acts as an ortho, para directing group but is deactivating. Use the Lewis structure of the nitroso group and its inductive and resonance...
-
Polystyrene (polyethenylbenzene) is a familiar polymer used in the manufacture of foam cups and packing beads. One could, in principle, synthesize polystyrene by cationic polymerization with acid....
-
Consider the following data for a clinical laboratory: a. Using ABC techniques, determine the cost allocation rate for each activity. b. Now, using these allocation rates, estimate the total cost of...
-
An analyst obtained predictions using exponential smoothing, see the table below. Year Month Sales ES forecast 2020 10 $1,450.00 $1,450.00 2020 11 $2,390.00 2020 12 $1,980.00 2021 1 $2,077.00 A 2021...
-
What laws and regulations were broken during the love canal and Hooker chemical incident in New York?
-
Last month McAlister Company had a $60,000 loss on sales of $300,000. Fixed costs are $120,000 a month. Answer the following questions: a. What was the contribution margin percentage? b. What monthly...
-
Why is receptor tyrosine kinase EGFR important for regulation of cell proliferation and cell survival?
-
In class we saw that Karatsuba multiplication allowed one to multiply two n-bit numbers in O(nlog(3)) time. It turns out that using the Fast Fourier Transform, one can multiply numbers in nearly...
-
Brown Inc., a calendar year taxpayer, reported the following transactions. Taxable income ................................................................................... $2,600,000 Depreciation...
-
After looking at the resources, explain what a spirit image is. Why might looking at a god and/or a human in terms of their spirit be helpful if you want to eliminate some of the divisions between...
-
A first -order decomposition reaction is observed to have the following rate constants at the indicated temperatures. Estimate the activation energy. k/(10-3 s-1) 2.46 45.1 576 0/C 20.0 40.0
-
Sucrose is readily hydrolyzed to glucose and fructose in acidic solution. The hydrolysis is often monitored by measuring the angle of rotation of plane polarized light passing through the solution....
-
Show that the following mechanism can account for the rate law of the reaction in Problem 22.11: What further tests could you apply to verify this mechanism? HCl + HCl K, HCI + CH,CH=CH, complex...
-
A taxpayer disposed a part of asset for 100,000 SR in cash. The taxpayer purchased that asset for 90,000 SR 5 years ago but the market value of the complete assets was as follows: Part disposed...
-
A company maintains debt/equity ratio of 0.6. The flotation cost of equity is 12% and flotation cost of debt it is 6%. The firm is considering a new project which will require $5 million in external...
-
Flight Caf prepares in-flight meals for airlines in its kitchen located next to a local airport. The company's planning budget for July appears below: Flight Caf Planning Budget For the Month Ended...

Study smarter with the SolutionInn App


