What alkene monomer gives the following polymer? CH3H C-C- H H
Question:
What alkene monomer gives the following polymer?
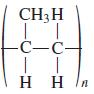
Transcribed Image Text:
CH3H C-C- H H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (9 reviews)
The structure ...View the full answer

Answered By

SUMAN MAHAPA
I am very keen in helping others to solve problems. I always try to teach in very simple & easy method, so that I can make teaching learning process easy and full of fun.Improvisation in study technique is my goal.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Identify? the structural class to which the following polymer belongs, and show the structure of the monomer units used to make it:
-
Consider the following polymer: Is this polymer a homopolymer or a copolymer, and is it formed by addition polymerization or condensation po-lymerization? What is (are) the monomer(s) for this...
-
Draw the structure of the monomer or monomers used to synthesize the following polymers: a. b. c. d. e. f. g. h. For each polymer, indicate whether it is a chain-growth polymer or a step-growth...
-
Discuss the advantages or disadvantages of subsidiary legislations in the Malaysian legal system in dealing with Covid-19 vaccination. Please write down the legal terms for discussion. Please do not...
-
1. Place yourself in Kevin's and Jeff's shoes. How do you respond to Reid concerning this delicate situation? Jeff and Kevin's first and only priority is the system automation projects, but if there...
-
What is the effect of the price floor, Pf, shown in the graph below, on price and quantity? Price P Supply Quantity Demand
-
In a contract dispute between a US company and a Canadian company, the contract itself referred to provisions of the Uniform Commercial Code. Do these references alone preempt the contract from being...
-
The purchases and issues of rubber gaskets (Materials Ledger #11216) as shown in the records of HD Corporation for the month of November follow: Required: 1. Complete a materials ledger account...
-
Direct labor-hours Machine-hours Total fixed manufacturing overhead cost Variable manufacturing overhead per machine-hour Variable manufacturing overhead per direct labor-hour Required: 1. Compute...
-
Jefferson Company is a merchandiser located in western North Carolina. You serve on the companys capital budgeting review committee whose purpose is to evaluate all capital investment proposals and...
-
Write out detailed step-by-step mechanisms for the reactions in parts (c), (e), (f), (h), (j), (k), (m), (n), (o), and (p) of Problem 46. (c) BH 3 , THF then NaOH + H 2 O 2 (e) HBr (f) HBr +...
-
Give the expected major product from reaction of 3-methyl-1-butene with each of the following reagents. Explain any differences in the products mechanistically. (a) 50% aqueous H 2 SO 4 ; (b) (c) BH...
-
Recalculate Dynamic Mattress?s 2019 financing plan (Table 19.6), assuming that the firm wishes to maintain a minimum cash balance of $30 million instead of $25 million. Assume the firm can convince...
-
How can an understanding of organizational behavior enhance the functions of managers in criminal justice organizations? Provide an example to substantiate your rationale.
-
Can get a summary of this book please The Next factory of the World: How Chinese investment is reshaping Africa by Irene Yuan Sun, and explanation about both China's role in the world and Africa's...
-
What do you understand are the 2 most important metrics in content marketing?Explain and justify your answer Which metrics are not related to content marketing and why? Mention at least 2
-
Critically assess how leadership style can affect the dynamics of a group. The Importance of leadership at organizational level An Assessment of different leadership styles followed by example of a...
-
What are the thoughts about work/life balance?Does it exist?Why or why not? What can companies do to offer work/life balance or something similar?
-
Natalie's estate includes the following assets. ________ ___________________________Fair Market Value______________ ____________________________Date of Death __________Six Months Later Apartment...
-
Using the information presented in Problem 13.4B, prepare a partial statement of cash flows for the current year, showing the computation of net cash flows from operating activities using the...
-
The equilibrium constant for the reaction N2 (g) + O,(g) ;:='02NO(g) is 1.69 x 10-3 at 2300 K. A mixture consisting of 5.0 g of nitrogen and 2.0 g of oxygen in a container of volume 1.0 dm3 is heated...
-
What is the standard enthalpy of a reaction for which the equilibrium constant is (a) doubled, (b) halved when the temperature is increased by 15 K
-
The dissociation vapour pressure ofNH4Cl at 427C is 608 kPa but at 459C it has risen to 1115 kPa. Calculate (a) The equilibrium constant, (b) The standard reaction Gibbs energy, (c) The standard...
-
The idea of a social contract is a powerful concept, but cannot be brought to life without active measures in society by all actors. South Africa can be argued to have a flawed version in place, but...
-
What is supplier economies and educational economies of scale in regards to external economies of scale? (section of my notes below, does not makes sense to me) Educational economies: local colleges...
-
Western Wholesale Foods incurs the following expenditures during the current fiscal year. Required: How should Western account for each of these expenditures? 1. Salaries for the repair technicians,...

Study smarter with the SolutionInn App


