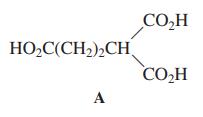
When acid A (margin) is heated to 230C, CO 2 and H 2 O are evolved and
Question:
When acid A (margin) is heated to 230°C, CO2 and H2O are evolved and a new compound is formed. Which one?
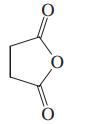
(a)
(b) HO2CCH2CH2CH2CH3
(c)
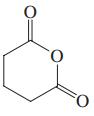
(d) CH3CH2CH(CO2H)2
(e)
Transcribed Image Text:
CO,H HO,C(CH2),CH CO,H А
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
A compound X contains 63.3 percent manganese (Mn) and 36.7 percent O by mass. When X is heated, oxygen gas is evolved and a new compound Y containing 72.0 percent Mn and 28.0 percent O is formed. (a)...
-
When compound A is heated, two isomeric products are formed. What are these two products? CO,H CO H Cl Compound A
-
You perform combustion analysis on a compound that contains only C and H. a. Considering the fact that the combustion products CO2 and H2O are colorless, how can you tell if some of the product got...
-
Carol Simon, the manager of Handy Plumbing has provided you with the following aging schedule for Handys accounts receivable: Carol indicates that the $125,200 of accounts receivable identified in...
-
"For my country, imports are the good thing about international trade, whereas exports are more like the necessary evil." Do you agree or disagree? Why?
-
Discuss the ways physicians and their practice behaviors affect the financial results of an operating budget and a capital budget.
-
When the plaintiff was 16, he was employed by Kmart as a cashier. At the end of his training, he was required to read Kmarts policy agreement, which included an agreement to submit all employment...
-
Depreciation ComputationReplacement, Nonmonetary Exchange Goldman Corporation bought a machine on June 1, 2008, for $31,800, f.o.b. the place of manufacture. Freight to the point where it was set up...
-
5. Consider uniform, inviscid, supersonic flow exhausting from a rocket engine nozzle. For fixed back pressure p (i.e., the ambient pressure outside the nozzle), qualitatively draw the first few wave...
-
Brain Hurt started a paper route on January 1, 1980. Every three months, he deposits $250 in his bank account, which earns 8 percent annually but is compounded quarterly. On December 31, 1984, he...
-
The reaction of ethyl butanoate with sodium ethoxide in CH 3 CH 2 OH gives (a) (b) (c) (d) CH;CH,CH,CHCHCO,CH,CH3 H,CH3
-
A compound with m.p. = -22C has a parent peak in its mass spectrum at m/z = 113. The 1 H NMR spectrum shows absorptions at = 1.2 (t, 3 H), 3.5 (s, 2 H), and 4.2 (q, 2 H). The IR spectrum exhibits...
-
Which theory discussed in the chapter would enable a court to hold Hanousek criminally liable for violating the statute regardless of whether he participated in, directed, or even knew about the...
-
Given the following information: Accounts receivable 31 December 2015 $281,962 Allowance for doubtful accounts balance: 31 December 2014 $3,693 Charged to costs and expenses: $2,951 Write-offs net of...
-
Brainstorm information about yourself that you want to include in your personal branding statement. > What type of position are you seeking? > What makes you qualified for this type of position? 2....
-
A computer system provides instructions with length 4 bytes each, and uses a page size of 1K bytes. Assume the following nonsense program. ORG READ LOOP MOVE SUB A B BLT ***** STOP ORG DS ... ORG DS...
-
Write a proposal on a Elon Musks star ship and why it would be good, who it would benefit, how much does it cost, and when it will get done?
-
Using the Corruption Perceptions Index on Transparency International's website, Transparency International 2012 Corruptions Perception Index, discuss how you would advise your senior management to...
-
Addison Parker, single and age 32, lives at 3218 Columbia Drive, Spokane, WA 99210. She is employed as regional sales manager by VITA Corporation, a manufacturer and distributor of vitamins and food...
-
A report from the college dean indicates that for the previous semester, the grade distribution for the Department of Psychology included 135 As, 158 Bs, 140 Cs, 94 Ds, and 53 Fs. Determine what kind...
-
Which, if any, of the four substances in Problem 2.43 is a strong enough acid to react almost completely with NaOH? (The pKa of H2O is 15.74.)
-
The ammonium ion (NH4+, pKa = 9.25) has a lower pKa then the methyl-ammonium ion (CH3NH3+, pKa = 10.66). Which is the stronger base, ammonia (NH3) or methylamine (CH3NH2)? Explain
-
Is tert-butoxide anion a strong enough base to react with water? In other words, can a solution of potassium tert-butoxide be prepared in water? The pKa of tert-butyl alcohol is approximately18. CH...
-
A bond currently sells at 92% of face value in the marketplace. The bond pays semi-annual coupons with a coupon rate of 4.2% APR. The face value of the bond is $1,000, and the bond will mature in...
-
1. Micky's auto mechanic shop has a daily total cost represented by: C 98+2q+2q2. Shop's marginal cost is given by: MC 2+ 4q, where q is the number of repaired cars. = a. Derive an expression for...
-
Look at the graphs below for Country A and Country B. Wine Wine Country A-PPF 40 Country B-PPF 10 10 40 Cheese 20 Cheese a. Which country likely has a comparative advantage in wine and which country...

Study smarter with the SolutionInn App



