Which of the following structures qualify as being aromatic, according to Hckels rule? CH=CH2 (a) A (b)
Question:
Which of the following structures qualify as being aromatic, according to Hückel’s rule?
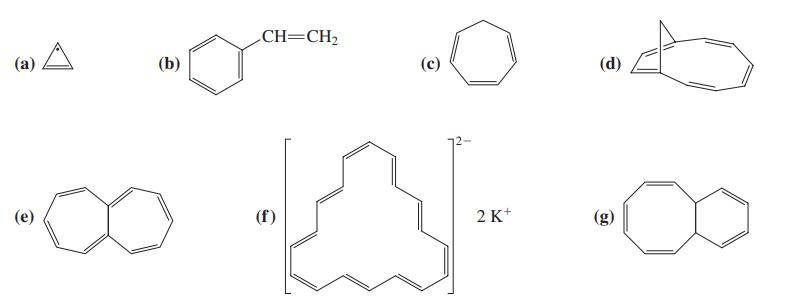
Transcribed Image Text:
CH=CH2 (a) A (b) (с) (d) "00 (е) (f) 2 K*
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (6 reviews)
For a compound to be aromatic according to Huckels rule it shoul...View the full answer

Answered By

Aditi Deswal
Currently , I am doing post graduation ( MSc.) in Chemistry . I want to bacome a lecturer . I feel happy when I teach student and solve their problem . It is my passion as well as hobby . I feel blessed If I got a chance to share my knowledge as much as I have . I teach student at my home also. I love teching and want to spend my whole life to teach the students and explores new things.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Which of the following structures has the smallest heat of combustion? (a) (b) (c) (d) CH3 CH3 CH3 CH3 CH3
-
Which of the following structures represent the same compound? Which ones represent different compounds? (a) (b) (c) (d) (e) (f) (g) Name the structures given in Problem 3-33, parts (a), (c), (e),...
-
Which of the following compounds are aromatic? Are any antiaromatic? NH HN: :NH Nl 1
-
Lamonda Corp. uses a job order cost system. On April 1, the accounts had the following balances: The following transactions occurred during April: (a) Purchased materials on account at a cost of...
-
Differentiate between the systems approach and the systems analysis approach taught in introductory systems analysis and design courses.
-
A not-for-profit organization uses any revenue to reward the management team. True or False
-
What would happen to the SML graph in Figure 8.8 if expected inflation increased or decreased? Figure 8.8 268 269 270 271 272 273 274 275 A Required Rate of Return TH-13.0% SML: r, RF+RPM * b D E F H...
-
Revenues, production, and purchases budgets. The Suzuki Co. in Japan has a division that manufactures two-wheel motorcycles. Its budgeted sales for Model G in 2010 are 900,000 units. Suzukis target...
-
A jewelry shop specializes in creating gold and silver charms. The shop has five metalsmiths who work on the charms. One week, the shop's production possibilities curve shifts inward. What could...
-
The Springfield School Board has made the decision to close one of its middle schools (sixth, seventh, and eighth grades) at the end of this school year and reassign all of next year's middle school...
-
Complete hydrogenation of 1,3,5,7-cyclooctatetraene is exothermic by -101 kcal mol -1 . Hydrogenation of cyclooctene proceeds with H = 223 kcal mol -1 . Are these data consistent with the description...
-
Following are spectroscopic and other data for several compounds. Propose a structure for each of them. (a) Molecular formula = C 6 H 4 Br 2 . 1 H NMR spectrum A. 13 C NMR: 3 peaks. IR: v = 745 (s,...
-
An accounting professor wants to know the average GPA of the students enrolled in her class. She looks up information on Blackboard about the students enrolled in her class and computes the average...
-
Two signals are spaced at 1,000 ft on an urban arterial. It is desired to establish the offset between these two signals, considering only the primary flow in one direction. The desired progression...
-
The Universal Orlando Resort is a theme park resort in Orlando, Florida. It is wholly owned by NBC Universal and its affiliates, and is the largest property operated by Universal Parks & Resorts....
-
Budgetary Oversight: A state is experiencing a budget deficit in one of its major funds. Discuss the role of budgetary accounting in identifying and controlling over-expenditures. How would you...
-
Tolton, Inc. is just shy of hitting its operating income target. The manager, K.T. Tolton, decides to purchase inferior materials right before year end. The standard price for the materials is $14.00...
-
Big Rock's revenue recognition is based on product sales at the time the product is shipped, and the following conditions exist: Title has passed to the purchaser according to the shipping terms. ...
-
A shareholder's basis in property received in a stock redemption is the same whether the redemption is qualifying or nonqualified. Comment on the validity of this statement?
-
Q1) What is the a3 Value Q2) What is the a7 Value Q3) What is the a4 Value Q4) What is the b3 Value Q5) What is the b2 Value Q6) What is the sign of 2nd constraint? A pastry chef at a bakery wants to...
-
The dipole moment of bromobenzene is 5. 17 X 10-30 C m and its polarizability volume is approximately 1.5 x 10-29 m3. Estimate its relative permittivity at 25C, when its density is 1491 kg m-3.
-
Calculate the vapour pressure of a spherical droplet of water of radius 20.0 nm at 35.0e. The vapour pressure of bulk water at that temperature is 5.623 kPa and its density is 994.0 kg m-1.
-
The contact angle for water on clean glass is close to zero. Calculate the surface tension of water at 30C given that at that temperature water climbs to a height of 9.11 cm in a clean glass...
-
Think back to a job situation in which your manager took action to reprimand, redirect, or reinforce your performance. 1. What did you do, and how was that behavior perceived by your manager? Was the...
-
Can you discuss the security features and mechanisms implemented in advanced operating systems to protect against various threats, including privilege escalation, malware, and denial-of-service...
-
Takeaways from the following each articles:- 1. "The Case for AI Insurance," by R. S. Siva Kumar and Frank Nagle, Harvard Business Review , April 29 2020. 2. "AI is Changing Work - and Leaders Need...

Study smarter with the SolutionInn App


