Which of the three compounds below would be the most reactive toward hydrolysis with aqueous base? (a)
Question:
Which of the three compounds below would be the most reactive toward hydrolysis with aqueous base?
(a)
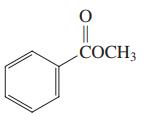
(b)
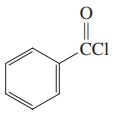
(c)
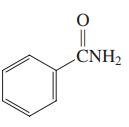
Transcribed Image Text:
СОСH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (7 reviews)
correct option is b Explanation These are all acid derivatives The reactivity of a...View the full answer

Answered By

Showket Ahmad khoja
I have Masters degree in chemistry and I am topper of my batch. I have also Qualified J&K SET . I am also winner of national Chemistry quiz. I have 5 years of experience in teaching Chemistry to 11th, 12th , NEET, JEE , +2 and +3 levels. I am both offline as well as online tutor. I am currently teaching Chemistry at CM'S Model Boys Higher secondary school Kupwara j&k India
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Which of the three primary composite geometries is most likely to possess isotropic properties?
-
Which compound is more reactive toward electrophilic substitution (for example, nitration)? OCH or b. CH,CH3 a. ETor
-
Which of the three basic philosophies of social responsibility would you find most appealing as the chief executive of a large corporation? Explain.
-
Line A: y = 3 0.6x Line B: y = 4 x a. Graph the linear equations and data points. b. Construct tables for x, y, Ëy, e, and e 2 similar to Table 4.4 on page 151. c. Determine which line fits the...
-
Looking at the last year of the data, compare the average openness of the 20 largest countries (measured in terms of population) and the 20 smallest countries. Which is more open?
-
Why is employee-centricity alone not enough?
-
Zappos.com is a popular website known mainly for its discounted shoe sales. In 2012, a hacker hacked into the Zappos website in an effort to obtain the personal account information of Zappos...
-
The following events apply to Montgomery Company for 2016, its first year of operation: 1. Received cash of $36,000 from the issue of common stock. 2. Performed $48,000 of services on account. 3....
-
1. The capital market is a market prepared for trading stocks, securities. The capital market acts as a liaison between investors and companies or government institutions through long-term trading...
-
Write a program that checks a text file for several formatting and punctuation matters. The program asks for the names of both an input file and an output file. It then copies all the text from the...
-
The best description for compound A (see margin) is (a) An amide; (b) A lactam; (c) An ether; (d) A lactone. H2C-C H2C-O A
-
Give the products of reaction of methyl pentanoate with each of the following reagents under the conditions shown. (a) NaOH, H 2 O, heat; then H + , H 2 O (b) (CH 3 ) 2 CHCH 2 CH 2 OH (excess) , H +...
-
Ally Cat LLC bought a condo from Chauvin, the seller of the unit. A Home Owners Limited Warranty document, containing an arbitration provision, was transferred to Ally Cat. However, the document did...
-
The next dividend payment by Savitz, Inc., will be $2.43 per share. The dividends are anticipated to maintain an annual growth rate of 3.5% forever. If the stock currently sells for $70 per share,...
-
Burke's Corner currently sells blue jeans and T-shirts. Management is considering adding fleece tops to its inventory to provide a cooler weather option. The tops would sell for $50 each with...
-
1. For each of the following lists, perform a selection sort and show the list after each exchange that has an effect on the list ordering: a. 4, 8, 2,6 b. 12, 3, 6, 8, 2, 5, 7 c. D, B, G, F, A, C,...
-
"What challenges and opportunities are associated with motivation measurement and assessment, considering factors such as the development of psychometrically sound instruments, cultural and...
-
Following is the portfolio held by Kamil: Stock Investment XT RM 40,000 XU 50,000 XV 70,000 XW 65,000 The beta value for Stock XT is 0.70, Stock XU is 0.64, Stock XV is 1.40 and Stock XW is 1.000....
-
Ross would like to dispose of some land he acquired five years ago because he believes that it will not continue to appreciate. Its value has increased by $50,000 over the five-year period. He also...
-
When the concentration of a strong acid is not substantially higher than 1.0 10-7 M, the ionization of water must be taken into account in the calculation of the solution's pH. (a) Derive an...
-
Show the products of these elimination reactions and indicate which ismajor: OTs . b) CH,OH + CH,0 + OH ELOH CI E:OH + CH,CH,O c)
-
The reaction of 2-bromobutane with ethoxide ion in ethanol gives 81% of a mixture of (Z)- and (E)-2-butene. Explain which stereo isomer you expect to predominate in this mixture.
-
Show the products of these reaction and indicate which ismajor: N(CH3)3 N(CH3)3 a) b) d) c) CH3 .
-
Write a program in Ruby to find all prime numbers within a given range using the Sieve of Eratosthenes algorithm.
-
Question 1 [CLO 2] For the following transactions pass journal entries, prepare ledger accounts and also prepare the trial balance. 2018 April 1. Saeed started business with cash $200,000, stock...
-
i have written an informational interview e-mail to female television host and meet her to get to learn out of her. After a meeting i wants to thank her for sharing her time and knowledge to me. how...

Study smarter with the SolutionInn App


