Would you expect addition of HCl to the double bond of 3-buten-2-one (shown in the margin) to
Question:
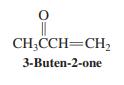
Would you expect addition of HCl to the double bond of 3-buten-2-one (shown in the margin) to follow Markovnikov’s rule? Explain your answer by a mechanistic argument.
Transcribed Image Text:
CH;CCH=CH, 3-Buten-2-one
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
Q Would you expect addition of HCl to the double bond ...View the full answer

Answered By

Seenaiah Gangipaka
I am post graguate(MSc-Organic Chemistry) and B.Ed(Physical Science).I have 11 years of good experience in teaching and professional.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
What two products are possible from the addition of HCl to 2-octene? Would you expect the reaction to be regiospecific?
-
What product(s) would you expect to obtain from reaction of 1, 3-cyclo-hexadiene with each of the following? (a) 1 mol Br2 in CH2C12 (b) O3 followed by Zn (c) 1 mol HCI in ether (d) 1 mol DCl in...
-
(a) How many stereoisomers are possible for 9, 10-dibromohexadecanoic acid? (b) The addition of bromine to palmitoleic acid yields primarily one set of enantiomers, ()-threo-9, 10-dibromohexadecanoic...
-
If a check correctly written and paid by the bank for $ 648 is incorrectly recorded on the company's books for $ 684 , the appropriate treatment on the bank reconciliation would be to THE ANSWER IS...
-
Matthew works as a paralegal for the prosecutor's office. While organizing a file in preparation for trial he comes across the name and telephone number of a witness who identified the perpetrator of...
-
Identify and explain the gender differences that exist in investing and gambling behavior.
-
In 2015, the city of San Francisco enacted an ordinance that required health warnings on advertisements for certain sugar-sweetened beverages (SSBs) that read: WARNING: Drinking beverages with added...
-
On November 1, 2013, Norwood borrows $ 200,000 cash from a bank by signing a five-year installment note bearing 8% interest. The note requires equal total payments each year on October 31. Required...
-
Suppose that farmers have a mandatory demand expansion program where all farmers pay a certain amount to fund a promotion program. You have estimated the following market supply and demand functions...
-
Santana Rey created Business Solutions on October 1, 2019. The company has been successful, and its list of customers has grown. To accommodate the growth, the accounting system is modified to set up...
-
Propose syntheses of the following compounds by using Michael additions followed by aldol condensations (i.e., Robinson annulation). Each of the compounds shown has been instrumental in one or more...
-
Using the following information, propose structures for each of these compounds. (a) C5H10O, NMR spectrum A, UV max (e) 5 280(18) nm; (b) C5H8O, NMR spectrum B, UV lmax(e) 5 220(13,200), 310(40) nm;...
-
To what extent would more knowledge of a specific individual change your answers? To what extent would more knowledge of a specific situation change your answers?
-
Cost of new computers $25,400 Salvage value of new computers at end of useful life $2,400 Life of new computers (years) 5 Market value of old computers today (equal to book value) $1,800 Annual...
-
A Venus-like planet has a 3400.0 km radius metallic core surrounded by a rocky mantle out to a total radius of 6770.0 km. The metallic core has a density of 15.8 g/cm3, and the rocky mantle has a...
-
Solving logarithmic equations with log terms on only one side of the equation: A. Make sure all log terms are on one side of the equation with non-log terms on the other side. B. Use the properties...
-
VOLMAX Fund is offering 5 mututal funds to the public. Volfgang, VOLMAX'S CIO, will sell to investors a risky portfolio WOLFX, which is made of the five basic funds. The Expected return, beta,...
-
As a college student who relies on a seamless internet connection for both home and campus network activities, identify the optimal internet package offered by an Internet Service Provider (ISP) that...
-
During the year, Rachel earned $18,000 of interest income on 2011 private activity bonds. She incurred interest expense of $7,000 in connection with amounts borrowed to purchase the bonds. What is...
-
What is master production scheduling and how is it done?
-
The transfer coefficient of a certain electrode in contact with M2+and M3+ in aqueous solution at 25C is 0.42. The current density is found to be 17.0 mA cm when the over voltage is 105 may. What is...
-
Determine the exchange current density from the information given in Exercise 25.16b.
-
Determine the effect that increasing the over potential from 0.50 V to 0.60 V has on the current density in the electrolysis of 1.0 M NaOH (aq), which is 1.22 mA cm-2 at 0.50 V and 25C. Take a= 0.50.
-
Given the following Python code: x = 20 while x > 10: #do something x = x 1 How many times this loop will iterate. 10 11 20 20 0
-
The HTML program is fine I want help to figure out what's wrong with CSS that would not make my website look the same as the picture I provided. @charset "UTF-8"; /* CSS Document for CA3 */ body {...
-
Consider a twisted pair link of distance 2 km. It is required to compute the amount of received power Pr, assuming the transmit power Pt = 1 Watt and the cable attenuation is 20 dB/km.

Study smarter with the SolutionInn App


