Write all products of the reaction of each of the alcohols in Problem 31 with concentrated H
Question:
Write all products of the reaction of each of the alcohols in Problem 31 with concentrated H2SO4 under elimination conditions.
In Problem 31
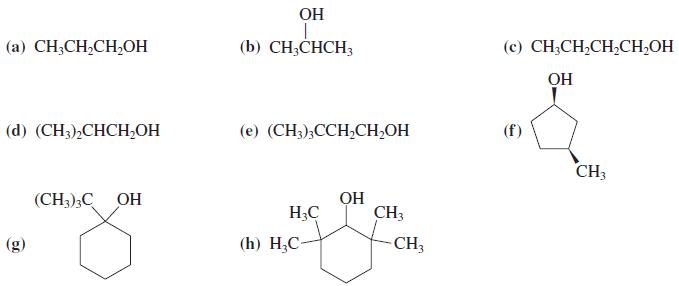
Transcribed Image Text:
OH (а) CH-CH-CH,Oн (b) CH;CHCH3 (c) CH;CH,CH,CH,OH OH (d) (CH),СНCН,ОН (е) (CH),ССH-CH-OH CH3 (CH3);C OH OH H3C CH3 (g) (h) H,С- CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
All alcohols when treated with concentrated Sulphuric acid under elimination ...View the full answer

Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Write all sensible products of the reaction of each of the alcohols in Problem 31 with concentrated aqueous HBr. In Problem 31 OH () CH-CH-CH,O (b) CH;CHCH3 (c) CH;CH,CH,CH,OH OH (d) (CH),C, ()...
-
Give the expected main product of the reaction of each of the alcohols in Problem 31 with PBr 3 . Compare the results with those of Problem 33.
-
Write the expected products of the reaction of each of the following sugars with (i) Br 2 , H 2 O; (ii) HNO 3 , H 2 O, 60C; (iii) NaBH 4 , CH 3 OH; and (iv) excess C 6 H 5 NHNH 2 , CH 3 CH 2 OH, ....
-
Suppose Targets stock has an expected return of 20% and a volatility of 40%, Hersheys stock has an expected return of 12% and a volatility of 30%, and these two stocks are uncorrelated. a. What is...
-
ist several of the team-building activities from the chapter and explain why they would be used during the execution phase.
-
Mynor Corporation manufactures and sells a seasonal product that has peak sales in the third quarter. The following information concerns operations for Year 2the coming yearand for the first two...
-
What is the plain-meaning rule?
-
William Bryant is the new owner of Ace Computer Services. At the end of August 2012, his first month of ownership, Bryant is trying to prepare monthly financial statements. Below is some information...
-
Arrow Hospitality prepares adjustments monthly and showed the following at September 3 0 , 2 0 2 3 : ARROW HOSPITALITY Trial Balances September 3 0 , 2 0 2 3 Unadjusted Adjusted Trial Balance...
-
The Pedal Pusher Bicycle Shop operates 7 days per week, closing only on Christmas Day. The shop pays $300 for a particular bicycle purchased from the manufacturer. The annual holding cost per bicycle...
-
Name the following alcohols according to the IUPAC nomenclature system. Indicate stereochemistry (if any) and label the hydroxy groups as primary, secondary, or tertiary. OH Br OH (a) CH;CH,CHCH; (b)...
-
Give detailed mechanisms and final products for the reaction of 3-methyl-2-pentanol with each of the reagents that follow. (a) NaH (b) Concentrated HBr (c) PBr 3 (d) SOCl 2 (e) Concentrated H 2 SO 4...
-
Use your statistical package to find the range, semi-interquartile range, unbiased variance, and unbiased standard deviation for each of the quantitative variables in Ihno's data set. (Does your...
-
Explain why swaps entail credit risk. Discuss the relationship between the value of the swap and credit risk exposure.
-
The standard portion size of sliced honey pork loin is 8 ounces, and the current edible portion cost is $0.30 an ounce. If the kitchen staff slices 10-ounce portions and the restaurant sells 50...
-
What innovative techniques can be employed to enhance data triangulation and validity in qualitative research designs?
-
How does Bourdieu's theory of social capital elucidate the ways in which social networks and cultural resources facilitate the acquisition and accumulation of symbolic power within a given social...
-
A museum curator tracked the ages of visitors who frequented different exhibits and displayed the data in a table. Dinosaurs Modern Art Inventions Aerospace Under 12 35 5 14 16 12-25 20 8 28 7 Over...
-
On January 1, 2012, Piranto acquires 90 percent of Slinton's outstanding shares. Financial information for these two companies for the years of 2012 and 2013 follows: Assume that a tax rate of 40...
-
Floyd Distributors, Inc., provides a variety of auto parts to small local garages. Floyd purchases parts from manufacturers according to the EOQ model and then ships the parts from a regional...
-
A Carnot cycle uses 1.00 mol of a monatomic perfect gas as the working substance from an initial state of 10.0 atm and 600 K. It expands isothermally to a pressure of 1.00 atm (Step 1), and then...
-
At 310 K, the partial vapour pressures of a substance B dissolved in a liquid A are as follows: xB 0.010 0.015 0.020 Pg/kPa 82.0 122.0 166.1 Show that the solution obeys Henry's law in this range of...
-
Discuss the implications for phase stability of the variation of chemical potential with temperature and pressure.
-
Access the IASB Web site IFRS - Resources for students. You can also use other sources to answer the following. a. Explain how the IFRS board develops IFRS Standards. b. Summarize the main features...
-
Bob's Bakery purchased an industrial oven for $22,000 with a residual value of $6,000 and a life expectancy of 8 years. Using straight-line depreciation, the amount of the depreciation adjustment for...
-
"2021 Fund is given to Groups whose 2021 Actual is more than or equal to the 2021 What If. The fund is $1000,000 plus $350 for each 2021 Actual number that is more than 2021 What If number, minus...

Study smarter with the SolutionInn App


