Write the products of each of the following reactions after aqueous work-up. (e) Write the results that
Question:
Write the products of each of the following reactions after aqueous work-up.
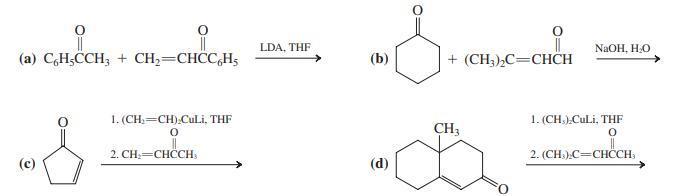
(e) Write the results that you expect from base treatment of the products of reactions (c) and (d).
Transcribed Image Text:
LDA, THE (a) CH;CCH; + CH;=CHCC,H; NaOH, HO + (CH;),C=CHCH 1. (CH,=CH).CULI, THF 1. (CH.).CuLi, THF CH3 2. CН— СНCСH 2. (CH:).C=CHCCH, (d)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (6 reviews)
Results from a wide range of climate model simulations ...View the full answer

Answered By

Dudhat Vaidehi
I tutored mostly elementary school students privately after school and during the summer. We met in their homes or at the public library. I charged an hourly fee, and I provided any necessary materials.
Having taught in special education in two local schools for many years meant that I had contact with a lot of parents of special needs students. I never had to advertise — word of mouth was how most folks knew of me. At one point I did have a website, but didn't utilize it much. I stayed very busy, especially in the summers, and always had a full schedule. I typically met with each student's teacher in order to get an idea of what the focus of my instruction/remediation should be. Becoming familiar with the student's learning style(s) was also very helpful. Often parents would share records and test results with me. After each tutoring session, I documented the student’s progress and gave parents written updates, as well as phone calls or emails as needed.
While my students and I certainly utilized technology and the internet often during our sessions, I never tutored online or for any tutoring company, so am not familiar with the curriculums or methods used in those settings.
Tutoring one on one was very enjoyable and rewarding. My students and I had fun, and grew quite fond of one another. The extra income was a bonus. I had to retire from tutoring due to a physically handicapping disease, and miss my students very much.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Give the products of each of the following reactions: a. b. c. d. e. f. g. h. i. j. HCI CH2CH3 1. CH3CH2MgBr 1. CH3CH2MgBr excesS CH,CH,COCH 2. H20 ot.cum 1. LiAIH4 NO 2. H20 catalytic Ht + CH...
-
Give the products of each of the following reactions: a. b. c. d. CH,-CH-CH-CH2 + CH3C-C= C-CCH, CH CH CH3 CHa CH3C CH-CH-CCH3
-
Predict the products of each of the following reactions. (a) (b) (c) (d) OH Cl pyridine OH (1) NaH (2) CH2l HBr OH HNOg, H2SO4 H3C
-
The following are selected transactions of Bridgeport Department Store Ltd. for the current year ended December 31. Bridgeport is a private company operating in the province of Manitoba where PST is...
-
Apply the North Carolina assault statute found in Section 4-5 to the following facts. a. Bobby is angry with his supervisor. He takes a gun to work, intending to scare his supervisor. He waves the...
-
Discuss how the caregiver role affects investing.
-
Its Intoxicating Inc., is a Pennsylvania corporation that manufactures and distributes cosmetic products to various retailers. Maritim is a German company that owns and operates hotels throughout...
-
The chart of accounts of LR Company includes the following selected accounts. 112 Accounts Receivable ......401 Sales Revenue 120 Inventory ...........412 Sales Returns and Allowances 126 Supplies...
-
Scenario 1: Joey is a 15 year old who is ready to work, but his parents want him to have more responsibility with money first. His parents have already set up checking and savings accounts for him at...
-
Vulcan Flyovers offers scenic over flights of Mount St. Helens, the volcano in Washington State that explosively erupted in 1982. Data concerning the company's operations in July appear below: The...
-
Give the expected product(s) of each of the following reactions. 1. LDA, THF 2. BICH,COCH, H 1. LDA, THF 2. CH.CH Br, HMPA (a) CH,CCH,CH,CH3 (b) 0:
-
Write the final products of the following reaction sequences. (a) (b) (c) (d) Write a detailed mechanism for reaction sequence (c). NaOCH, CH,O, O + CH2=CHCCH3
-
Use the first thirteen rules of inference to derive the conclusions of the following symbolized arguments: 1. A. (F.L) 2. A (UvW) 3. F (UvX) /Uv (W.X)
-
Total number of shares in Dhofar steel company are 10000 and each one is sold for RO 10. Mr. Ahmed invested RO 10000 in purchasing shares of the above company. Based on one share one vote, how much %...
-
Selling price for 100. Cash flows over 10 years 1 $12 2 $11 3 $13 4 $14.1 5 $12. 6 $13.3 7 $12 8 $13 9 $11 10 $10 The cash flow yield is closest to what percent?
-
Suppose a different company offers Ludo Corporation a "Loss Control" service which costs S (in dollars) and which cuts the probability of all losses in half (such that the probability of both the...
-
How much equity would Phil have in his home based on the following information: Purchased price of $425,000 Fair market value of $432,000 20% down payment Has made $15,000 in principal payments
-
Should civility be a criterion for job performance? Discuss the implications of leaders and managers who lack appropriate leadership skills.
-
Comment on the validity of the following statement: Because the regular tax NOL deduction is added back to the taxable income starting point of the corporate AMTI calculation, an NOL deduction is not...
-
Using Gauss-Jordan elimination, invert this matrix ONLY 0 0 0 0 1
-
The chemisorptions of hydrogen on manganese is activated, but only weakly so. Careful measurements have shown that it precedes 35 per cent faster at 1000 K than at 600 K. What is the activation...
-
The adsorption of a gas is described by the Langmuir isotherm with K = 0.777 kPa-1 at 25C. Calculate the pressure at which the fractional surface coverage is (a) 0.20, (b) 0.75.
-
A certain solid sample adsorbs 0.63 mg of CO when the pressure of the gas is 36.0 kPa and the temperature is 300 K. The mass of gas adsorbed when the pressure is 4.0 kPa and the temperature is 300 K...
-
The Foundational 15 (Algo) [LO1-1, LO1-2, LO1-3, LO1-4, LO1-5, LO1-6] [The following information applies to the questions displayed below.] Martinez Company's relevant range of production is 7,500...
-
Taylor Swift's most recent tour was a success by every measure. Named after the bestselling album of her career 1989, it grossed more than $250 million worldwide the top tour of 2015 raved about the...
-
Use the following to answer questions 34-36 The year-end adjusted trial balance of the Corporation included the following account balances: Retained earnings $200,000 Service revenue 600,000 Salaries...

Study smarter with the SolutionInn App


