The total assets and total liabilities for a recent year of Apple (AAPL) and HP (HPQ) formerly
Question:
The total assets and total liabilities for a recent year of Apple (AAPL) and HP (HPQ) formerly Hewlett-Packard are shown here.
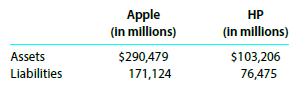
Determine the stockholders’ equity of each company.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
ANSWER To determine the stockholders equity of each co...View the full answer

Answered By

Aketch Cindy Sunday
I am a certified tutor with over two years of experience tutoring . I have a passion for helping students learn and grow, and I firmly believe that every student has the potential to be successful. I have a wide range of experience working with students of all ages and abilities, and I am confident that I can help students succeed in school.
I have experience working with students who have a wide range of abilities. I have also worked with gifted and talented students, and I am familiar with a variety of enrichment and acceleration strategies.
I am a patient and supportive tutor who is dedicated to helping my students reach their full potential. Thank you for your time and consideration.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Business questions
-
The total assets and total liabilities for a recent year of Best Buy and Gamestop are shown Determine the stockholders equity of eachcompany. Best Buy (in millions) $16,005 12,260 Gamestop (in...
-
The total assets and total liabilities for a recent year of Apple and Dell are shown here. Determine the stockholders equity of eachcompany. Apple (in millions) $176,064 57,854 Dell (in millions)...
-
The total assets and total liabilities for a recent year of Best Buy (BBY) and Gamestop (GME) are shown below. Determine the stockholders equity of each company. Best Buy (in millions) Gamestop (in...
-
Trade promotions refer to offers made to Group of answer choices channel members non-profit organizations businesses consumers Question 2 Consumer promotions refer to offers made to Group of answer...
-
Establishing successful interorganizational relationships is difficult, according to authorities. Will the success record improve in the future as more companies pursue this strategy?
-
A 0.3-m3 rigid tank is filled with saturated liquid water at 200°C. A valve at the bottom of the tank is opened, and liquid is withdrawn from the tank. Heat is transferred to the water such that...
-
Let \(V_{k s}\) be the value of an adjustable-rate loan initiated at period \(k\) and state \(s\) with initial principal of 100 . The loan is to be fully paid at period \(n\). The interest rate...
-
A machine with a book value of $49,000 has an estimated five-year life. A proposal is offered to sell the old machine for $30,000 and replace it with a new machine at a cost of $64,000. The new...
-
Manufacturing costs for Davenport Company during 2018 were as follows: Beginning Finished Goods, January 1 Beginning Raw Materials, January 1 Beginning Work in Process, January 1 Direct Labor for the...
-
Assume that a friend of yours operates a family-owned pharmacy. A super Wal-Mart, scheduled to open in the next several months, will also offer pharmacy services. What business emphasis would your...
-
From the following list of selected items taken from the records of Flip Flop Sandals Inc. as of a specific date, identify those that would appear on the balance sheet. 1. Accounts Receivable 2....
-
Given the joint density Show that µY|x = 1 + 1/x and that var(Y|x) does not exist. 2r for x0 and y >0 elsewhere
-
A benzene pipeline that is 100 ft and runs horizontally arises a leak 43 feet from the high- pressure end. It is thought that the size of the leak is 0.1 inch. The pressure at the beginning of the...
-
Calculate the frequency of light with a wavelength of 6.0 nm. 14 -1 O a. 5 x 10 s 15 -1 O b. 5 x 10 S O c. 5 x 10 O d. 5 x 10 16-1 S 17 -1 S
-
Earth's primitive atmosphere, which consisted mainly of H20 vapor and CO2, formed by a process called Acid reflux Outgassing Vaporization Condensation
-
Would a polar protic solvent like ethanol favor the solvolysis of 2-chloro-2-methylpropane? Why or why not? View Available Hint(s) O Ethanol would favor the solvolysis; the mechanism is E1. O Ethanol...
-
Prepare a 0.15% (w/v) solution of haemoglobin in 10 mM HCl pH2. Dilute to 0.03, 0.05, 0.07% and 0.09% in 10 mM HCl pH 2. 2. Prepare a calibration curve of haemoglobin in 10mM HCl pH 2 at 372 nm...
-
A loan of $6000 was repaid by quarterly payments of $450. If interest was 12% compounded monthly, how long did it take to pay back the loan?
-
What are technical skills At what level are they most important and why?
-
Miller Co., which produces and sells skiing equipment, is financed as follows: Bonds payable, 10% (issued at face amount) $10,000,000 Preferred $1 stock, $10 par 10,000,000 Common stock, $25 par...
-
Grodski Co. produces and distributes semiconductors for use by computer manufacturers. Grodski Co. issued $24,000,000 of 20-year, 10% bonds on April 1 of the current year, with interest payable on...
-
On the first day of its fiscal year, Robbins Company issued $50,000,000 of five-year, 8% bonds to finance its operations of producing and selling home improvement products. Interest is payable...
-
The local theater company is choosing its next play. The company's members recently held a vote to decide which of three choices - Elektra, Tartuffe, and Hamlet - the company will perform. The...
-
what are the benefits of the relational model relationships and ERD ( entity-relationship model)?
-
1.Libby just expanded her restaurant. She projects revenue will reach $35,000 for the new restaurant in the first year and increase by 25% over the next three years. Expenses are 75% of sales. The...

Study smarter with the SolutionInn App


