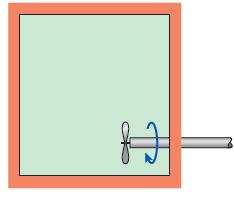
A substance is contained in a well-insulated rigid container that is equipped with a stirring device, as
Question:
A substance is contained in a well-insulated rigid container that is equipped with a stirring device, as shown in Fig. P4–30. Determine the change in the internal energy of this substance when 15 kJ of work is applied to the stirring device.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (5 reviews)
A substance is contained in a wellinsulated rigid container tha...View the full answer

Answered By

Fahmin Arakkal
Tutoring and Contributing expert question and answers to teachers and students.
Primarily oversees the Heat and Mass Transfer contents presented on websites and blogs.
Responsible for Creating, Editing, Updating all contents related Chemical Engineering in
latex language
4.40+
8+ Reviews
22+ Question Solved
Related Book For 

Thermodynamics An Engineering Approach
ISBN: 9781259822674
9th Edition
Authors: Yunus Cengel, Michael Boles, Mehmet Kanoglu
Question Posted:
Students also viewed these Engineering questions
-
A 15 L tank contains 1 kg of R-12 refrigerant at 100oC. It is heated until the temperature of the refrigerant reaches 150oC. Determine the change in. (a) Internal energy (U). (b) Entropy, (S). Use...
-
A rigid container equipped with a stirring device contains 2.5 kg of motor oil. Determine the rate of specific energy increase when heat is transferred to the oil at a rate of 1 W, and 1.5 W of power...
-
In a refrigerator, 2.00 mol of an ideal monatomic gas are taken through the cycle shown in the figure. The temperature at point A is 800.0 K. (a) What are the temperature and pressure at point D? (b)...
-
Discuss why marketing management is important to an organization, and how corporate and business strategy analysis gives an organization a competitive advantage.
-
The owner of a restaurant serving Continental-style entrees has the business objective of learning more about the patterns of patron demand during the Friday-to-Sunday weekend time period. Data were...
-
For Exercises use the traditional method of hypothesis testing unless otherwise specified. Assume the assumptions have been met. Random samples of summer gasoline prices per gallon are listed for...
-
Explain why employers should place disclaimers in employee handbooks.
-
A car dealer acquires a used car for $ 14,000, terms FOB shipping point. Additional costs in obtaining and offering the car for sale include $ 250 for transportation-in, $ 900 for import duties, $...
-
Overview One sector that has witnessed significant changes during the last two years is the grocery sector. Supermarkets and firms had to be creative in ensuring that consumers continued to get...
-
1. What are some advantages of a person-focused pay system at Mitron? 2. What are some disadvantages? 3. What approach would you recommend for Holly to take in designing a person-focused pay system?...
-
Complete each line of the following table on the basis of the conservation of energy principle for a closed system. W. E, E, e2- e; kJ/kg m out kJ kJ kJ kJ kg 280 1020 550 860 3 5 -350 130 260 -150...
-
A rigid 1-ft 3 vessel contains R-134a originally at 20F and 27.7 percent quality. The refrigerant is then heated until its temperature is 100F. Calculate the heat transfer required to do this. Heat...
-
For the equilibrium Kp = 8.5 Ã 10-3 at 150 oC. If 0.025 atm of IBr is placed in a 2.0-L container, what is the partial pressure of all substances after equilibrium is reached? 2 IBr(g)12(g)...
-
What is the benchmark for each ratio? Why is it important to use benchmarks or historical numbers for comparison? Are Marabelle's ratios falling within appropriate range? Explain. What elements does...
-
Calculate the adjusted cost base of the shares: Option price: $15 per share FMV of the shares when the options were exercised: $17 per share Bargain element: $15 - $17 = -$2 per share Number of...
-
What are the different formats available for presenting the income statement? What elements does it show that are important for decision making?
-
Both Medical Manufacturing and R&D Health Inc. have developed advanced hearing aid technologies. R&D's hearing aids sell at $4,000 per pair, whereas Medical Manufacturing sells its hearing aids at...
-
How do you distinguish between the concept of development economics and that of traditional economic growth? Do you agree with the idea of many economists who maintain that development economics has...
-
Shoppers Drug Mart's biggest competitor, after Jean Coutu, is the Katz Group. Its Canadian stores include Rexall Drug Stores, Guardian, Medicine Shoppe, and Pharma Plus Drugmart, among others. Katz...
-
An interest bearing promissory note for 90 days at 5.6% p.a. has a face value of $120,000. If the note is discounted 20 days after the issue date at a rate of 6.8% p.a., calculate the amount of...
-
Saturated steam coming off the turbine of a steam power plant at 40oC condenses on the outside of a 3-cmouter- diameter, 35-m-long tube at a rate of 130 kg/h. Determine the rate of heat transfer from...
-
Water in a 5 - cm-deep pan is observed to boil at 98oC. At what temperature will the water in a 40 - cm-deep pan boil? Assume both pans are full of water.
-
A cooking pan whose inner diameter is 20 cm is filled with water and covered with a 4-kg lid. If the local atmospheric pressure is 101 kPa, determine the temperature at which the water starts boiling...
-
Required Information [The following Information applies to the questions displayed below.] Simon Company's year-end balance sheets follow. At December 31 Assets Current Year 1 Year Ago 2 Years Ago...
-
Superior Company provided the following data for the year ended December 31 (all raw materials are used in production as direct materials): Selling expenses Purchases of raw materials Direct labor...
-
Correlle dishes are known for being mostly unbreakable in daily use. Kevin's 12-year-old son decided to test this "unbreakable" idea by throwing a plate hard onto the floor. It broke. what this is an...

Study smarter with the SolutionInn App


