Determine the total heat transfer for the reversible process 1-3 shown in Fig. P758. 3 360 55
Question:
Determine the total heat transfer for the reversible process 1-3 shown in Fig. P7–58.
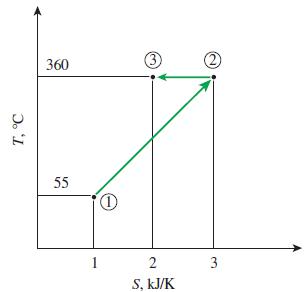
Transcribed Image Text:
3 360 55 1 2 S, kJ/K T, °C
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (14 reviews)
The total heat transfer for the process 13 shown ...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Thermodynamics An Engineering Approach
ISBN: 9781259822674
9th Edition
Authors: Yunus Cengel, Michael Boles, Mehmet Kanoglu
Question Posted:
Students also viewed these Engineering questions
-
Determine the total heat transfer for the reversible process 1-2 shown in Fig. P759. 500 100 0.2 1.0 S, kJ/K T, C
-
A cylinder fitted with a spring-loaded piston serves as the supply of steam for a steam turbine. Initially, the cylinder pressure is 2 MPa and the volume is 1.0 m3. The force exerted by the spring is...
-
An air duct heater consists of an aligned array of electrical heating elements in which the longitudinal and transverse pitches are SL = ST = 24 mm. There are 3 rows of elements in the flow direction...
-
In Problems 1316, three matrices A, B, and C are given. Verify by computation of both sides the associative law A(BC) = (AB)C. 25 = [ 3 i] c = [_ [-] -3 1 A = [2 -1], B=
-
Suppose the current stock price is $100, the exercise price is $100, the annually compounded interest rate is 5 percent, the stock pays a $1 dividend in the next instant, and the quoted call price is...
-
Assume that the weight of cereal in a 10-ounce box is N(, 2 ). To test H 0 : = 10.1 against H 1 : > 10.1, we take a random sample of size n = 16 and observe that x = 10.4 and s = 0.4. (a) Do we...
-
Confirm that the definition of the streamfunction given in the text satisfies the continuity equation. Use this in the N-S equation and derive the form for the \(E^{4}\) operator in spherical...
-
Consider the piston ring data shown in Table 6.3. Assume that the specifications on this component are 74.000 ï±ï 0.05 mm. (a) Set up x and R control charts on this process. Is the...
-
On January 1, 2024, Kroll Corporation paid $2,559,000 for 31 percent of the outstanding voting stock of Sharon, Incorporated, and appropriately applied the equity method for its investment. Any...
-
In the potential outcomes framework with heterogeneous (nonconstant) treatment effect, write the error as u i = (1 x i )u i (0) + x i u i (1). Let s 2 0 = Var[u i (0)] and s 2 1 Var[ui(1)]. Assume...
-
Water at 10C and 81.4 percent quality is compressed isentropically in a closed system to 3 MPa. How much work does this process require in kJ/kg?
-
Calculate the heat transfer, in kJ/kg. for the reversible steady-flow process 1-3 shown on a T-s diagram in Fig. P760. 120 100 30 0.02 1.0 s, kJ/kg-K T, C
-
Transmitting too much information for the receiver to process adequately results in ______ ______.
-
Brad and Rochelle agree to form Sail Away, Inc., a small family boat rental business. Brad agrees to contribute 20 sailboats that he bought over many years (and had a lot of fun with!) with a total...
-
When a given elastic band is stretched 0.37 m it has 1.32 J of elastic potential energy stored in it. What is the force constant for the elastic band?
-
How much energy is consumed with running a 60W light for 6 hour and 15min?
-
Marketing Strategy portion of the plan (55) o A discussion on the new environmental running shoes you would introduce to Adidas portfolio as their marketing manager. (5) o The growth strategy that...
-
1. What are some of the differences between the metric system and the standard system? 2. Why is the metric system the preferred method of measurement in the world today? 3. In detail, explain why or...
-
In 2013, Muhammad purchased a new computer for $16,000. The computer is used 100% for business. Muhammad did not make a 179 election with respect to the computer. He elects not to take additional...
-
President Lee Coone has asked you to continue planning for an integrated corporate NDAS network. Ultimately, this network will link all the offices with the Tampa head office and become the...
-
It is often stated that the refrigerator door should be opened as few times as possible for the shortest duration of time to save energy. Consider a household refrigerator whose interior volume is...
-
An air-conditioning system is used to maintain a house at a constant temperature of 20C. The house is gaining heat from outdoors at a rate of 20,000 kJ/h, and the heat generated in the house from the...
-
A Carnot heat pump is used to heat and maintain a residential building at 75F. An energy analysis of the house reveals that it loses heat at a rate of 2500 Btu/h per F temperature difference between...
-
Let's assume your lab balloons, when filled with air, each had a mass of 3.00 grams. In a variation of your lab activity, you attach one of these balloons to a string such that the distance from the...
-
Samantha normally requires 1 3 7 0 0 kJ ( about 3 2 7 4 Calories ) of food energy per day. If Samantha consumes 1 4 3 8 5 kJ per day, she will steadily gain weight. How much time must Samantha spend...
-
Five resistors with known resistances R = 5.00 2, R2 = 5.00 2, R3 = 3.00 Q2, R4 = 5.00 Q, and R5 = 5.00 Q are connected to a battery with emf = 7.00 V as shown in the figure. Please answer in Sl...

Study smarter with the SolutionInn App


