Methane (CH 4 ) is burned with 300 percent excess air in an adiabatic constant-volume container. Initially,
Question:
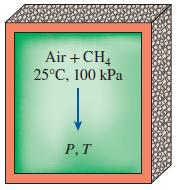
Methane (CH4) is burned with 300 percent excess air in an adiabatic constant-volume container. Initially, air and methane are at 1 atm and 25°C. Assuming complete combustion, determine the final pressure and temperature of the combustion products.
Transcribed Image Text:
Air +CH, 25°C, 100 kPa P,T
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 81% (11 reviews)
Methane is burned with 300 percent excess air adiabatically in a constant volume container The final ...View the full answer

Answered By

ANDREW KIPRUTO
Academic Writing Expert
I have over 7 years of research and application experience. I am trained and licensed to provide expertise in IT information, computer sciences related topics and other units like chemistry, Business, law, biology, biochemistry, and genetics. I'm a network and IT admin with +8 years of experience in all kind of environments.
I can help you in the following areas:
Networking
- Ethernet, Wireless Airmax and 802.11, fiber networks on GPON/GEPON and WDM
- Protocols and IP Services: VLANs, LACP, ACLs, VPNs, OSPF, BGP, RADIUS, PPPoE, DNS, Proxies, SNMP
- Vendors: MikroTik, Ubiquiti, Cisco, Juniper, HP, Dell, DrayTek, SMC, Zyxel, Furukawa Electric, and many more
- Monitoring Systems: PRTG, Zabbix, Whatsup Gold, TheDude, RRDtoo
Always available for new projects! Contact me for any inquiries
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Thermodynamics An Engineering Approach
ISBN: 9781259822674
9th Edition
Authors: Yunus Cengel, Michael Boles, Mehmet Kanoglu
Question Posted:
Students also viewed these Engineering questions
-
Methane (CH4) is burned with 200 percent excess air in an adiabatic constant volume container. Initially, air and methane are at 1 atm and 25oC. Assuming complete combustion, determine the final...
-
Methane fuel (CH4) is burned with 15 percent excess air in a space-heating furnace. The pressure in the chimney is 1 atm. Presuming complete combustion, determine the temperature of the combustion...
-
Ethyl alcohol [C 2 H 5 OH(g)] is burned with 200 percent excess air in an adiabatic, constant-volume container. Initially, air and ethyl alcohol are at 100 kPa and 25C. Assuming complete combustion,...
-
This bar chart displays the demographics (age group and gender) of a Business Analysis class Business Analysts Students 23-33 3410 M lem How many male students are in the class? 65 80 130 50
-
In a survey, the Canadian Automobile Association (CAA) found that 6.1% of its members bought their cars at a used-car lot. If 15 CAA members are selected at random, what is the probability that 4 of...
-
A driven \(R L C\) circuit has \(V_{\mathrm{C}}=5.0 \mathrm{~V}, V_{\mathrm{R}}=7.0 \mathrm{~V}\), and \(V_{\mathrm{L}}=9.0 \mathrm{~V}\). The driving frequency is A. Higher than the resonance...
-
Estimate the magnitude of the magnetic field at location 2 in Figure P27.14 if the magnetic field magnitude at location 1 is \(0.27 \mathrm{~T}\). Note that real magnetic field lines spread out in...
-
Kendra Knight took part in a friendly game of touch football. She had played before and was familiar with football. Michael Jewett was on her team. In the course of play, Michael bumped into Kendra...
-
1. An unpolarized light beam is incident on the polarizes of a polarization experiment and the intensity of light beam emerging from the analyzer is measured as 100 Lumens. Now, if the analyzer is...
-
a. Prepare a cross tabulation of the data on Industry (rows) and Brand Revenue ($ billions). Use class intervals of 25 starting at 0 for Brand Revenue ($ billions). b. Prepare a frequency...
-
A fuel at 25C is burned in a well-insulated steady flow combustion chamber with air that is also at 25C. Under what conditions will the adiabatic flame temperature of the combustion process be a...
-
A large railroad has experimented with burning powdered coal in a gas turbine combustor. Fifty percent excess air was introduced to the combustor at 1380 kPa and 127C while the powdered coal was...
-
Evaluate the definite integral. Use a graphing utility to verify your result. n/3 -/3 4 sec 0 tane de
-
What were the effects of excluding a health insurance provision from the Social Security Act? How could we have changed our future?
-
In the story "A Devoted Son" by Anita Desai, Rakesh, the son, touches his father Varma's feet. What is the significance of this, and how does it change as the story progresses?
-
9) When a beam of light originally traveling in air, enters a piece of glass NORMALLY, its wavelength A) increases by a factor B) is reduced by a factor C) is unaffected.
-
After being struck by a bowling ball, a 1.4 kg bowling pin sliding to the right at 3.7 m/s collides head-on with another 1.4 kg bowling pin initially at rest. Find the final velocity of the second...
-
13. The Apple stock's price is $112.92 on 8/1/15 and becomes $110.30 on 9/1/15. In August, Apple gives a dividend of $0.52 per share. What is the holding period monthly return for Apple in August? A....
-
If you were hired to advise Michael Jeffries on A&F's current organizational design, what weaknesses and potential threats would you identify? What strengths and opportunities?
-
Hardin Services Co. experienced the following events in 2016: 1. Provided services on account. 2. Collected cash for accounts receivable. 3. Attempted to collect an account and, when unsuccessful,...
-
Propane (C3H8) is burned with 150 percent theoretical air. The air-fuel mass ratio for this combustion process is (a) 5.3 (b) 10.5 (c) 15.7 (d) 23.4 (e) 39.3
-
One kmol of methane (CH4) is burned with an unknown amount of air during a combustion process. If the combustion is complete and there are 1 kmol of free O2 in the products, the air-fuel mass ratio...
-
An equimolar mixture of carbon dioxide and water vapor at 1 atm and 60oC enter a dehumidifying section where the entire water vapor is condensed and removed from the mixture, and the carbon dioxide...
-
Greenwood Company manufactures two products-15,000 units of Product Y and 7,000 units of Product Z. The company uses a plantwide overhead rate based on direct labor-hours. It is considering...
-
1. In his job, Martin is handed several bricks. He carries them across a yard and hands them to a bricklayer. He thinks he works hard, but his sister tells him that he does not work on the bricks....
-
12-19. A train starts from rest at station A and accelerates at 0.5 m/s for 60 s. Afterwards it travels with a constant velocity for 15 min. It then decelerates at 1 m/s until it is brought to rest...

Study smarter with the SolutionInn App


