On a summer day in New Orleans, Louisiana, the pressure is 1 atm: the temperature is 32C;
Question:
On a summer day in New Orleans, Louisiana, the pressure is 1 atm: the temperature is 32°C; and the relative humidity is 95 percent. This air is to be conditioned to 24°C and 60 percent relative humidity. Determine the amount of cooling, in kJ, required and water removed, in kg, per 1000 m3 of dry air processed at the entrance to the system.
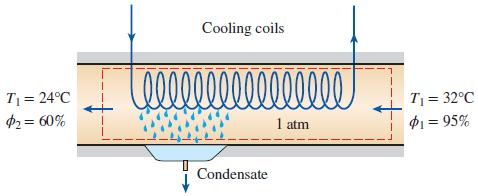
Cooling coils 00000000 T = 24°C $2= 60% T = 32°C P1 = 95% 1 atm Condensate
Step by Step Answer:
Air is cooled and dehumidified at constant pressure The amount of water removed from the air and the ...View the full answer



Thermodynamics An Engineering Approach
ISBN: 9781259822674
9th Edition
Authors: Yunus Cengel, Michael Boles, Mehmet Kanoglu
Related Video
In this video, the concept of pressure has been explained. The bottle is filled up with water, remove the pipe, and now you can see the clear flow of water from the holes due to liquid pressure.You can see that the second layer is holding the weight of the first layer and the third layer holding the weight of both upper layers, that’s why liquid flows out wider from the third as compared to the first two.At the first hole, water doesn’t roll along the walls; it flows out at an angle.At the second hole, you’ll see that the water flows out at a wider angle as compared to the first hole.At the third hole, we can see that the flow of water is much wider/greater than the other holes.This proves that the pressure at the bottom of the bottle is much more than the pressure at the top layers. P = ????gh
Students also viewed these Engineering questions
-
Reconsider Prob. 1482. How far will the temperature of the humid air have to be reduced to produce the desired dehumidification? Data From Reconsider Prob. 1482: On a summer day in New Orleans,...
-
During a summer day in El Paso, Texas, outdoor air is at 1 atm, 40C, and 20 percent relative humidity. Water at 20C is evaporated into this air to produce air at 25C and 80 percent relative humidity....
-
During a summer day in Phoenix, Arizona, the air is at 1 atm, 110oF, and 15 percent relative humidity. Water at 70oF is evaporated into this air to produce air at 75oF and 80 percent relative...
-
The following items were shown on the balance sheet of Herman Corporation on December 31, 2010: Stockholders Equity Paid-In Capital Capital Stock Common stock , $5 par value, 360,000 shares...
-
An observed frequency distribution is as follows: Number of successes 0 1 2 3 Frequency 89 133 52 26 a. Assuming a binomial distribution with n = 3 and p = 1/3, use the binomial probability...
-
Train A, \(m_{\mathrm{A}}=150,000 \mathrm{~kg}\), is traveling west at \(60 \mathrm{~km} / \mathrm{h}\). Train B, \(m_{\mathrm{B}}=100,000 \mathrm{~kg}\), behind train A on the same track, is...
-
There are about \(10^{11}\) stars in the Galaxy with mass comparable to the Sun. Determine total mass \(M_{S}\) in the stars and the fraction of the Galaxy mass in the form of interstellar medium...
-
The balance sheet for Garcon Inc. at the end of the current fiscal year indicated the following: Bonds payable, 8% (issued in 2004, due in 2024) ... $5,000,000 Preferred $4 stock, $50 par...............
-
QUESTION FOUR The increasing demand in risk management on the part of organizations has been identified as a general societal trend emphasizing public accountability and responsibility. State and...
-
1. Do you think this was an effective strategy for Pepsi? Do you think this resonated with the Millennial generation? 2. Do you think this is a good example of corporate social responsibility (CSR)?...
-
Atmospheric air from the inside of an automobile enters the evaporator section of the air conditioner at 1 atm, 27C, and 50 percent relative humidity. The air returns to the automobile at 10C and 90...
-
Repeat Prob. 1479 for a total pressure of 88 kPa for air. Data From Repeat Prob. 1479: Air enters a 40-cm-diameter cooling section at 1 atm, 32C, and 70 percent relative humidity at 120 m/min. The...
-
In general, what is one-way analysis of variance used for?
-
Determine the number of moles present in 98.6 g of nitric acid.
-
If a chemical reaction has a positive AH and a positive AS, then A) it will be spontaneous at all temperatures. B) it will be non-spontaneous at all temperatures. C) it will be spontaneous at high...
-
How many calories would be needed to boil 2.5 gal of water that was at 24.0 degrees C to start with?
-
2.78 To which carbon atoms in a heptane molecule can each of the following alkyl groups be attached without extending the longest carbon chain beyond seven carbons? b. Isopropyl c. sec-Butyl a. Ethyl...
-
How many atoms of tin are in a 36.5 g sample? Calculate the molar mass of the explosive TNT, C6HCH3 (NO2)3. Calculate the molar mass of phosphoric acid.
-
Roundtree Chocolates manufactures and distributes chocolate products. It purchases cocoa beans and processes them into two intermediate products: Chocolate powder liquor base. Milk chocolate liquor...
-
Using (1) or (2), find L(f) if f(t) if equals: t cos 4t
-
An ideal-gas mixture consists of 2 kmol of N2 and 6 kmol of CO2. The mass fraction of CO2 in the mixture is (a) 0.175 (b) 0.250 (c) 0.500 (d) 0.750 (e) 0.875
-
An ideal-gas mixture consists of 2 kmol of N2 and 4 kmol of CO2. The apparent gas constant of the mixture is (a) 0.215kJ/kgK (b) 0.225kJ/kgK (c) 0.243kJ/kgK (d) 0.875kJ/kgK (e) 1.24kJ/kgK
-
A rigid tank is divided into two compartments by a partition. One compartment contains 3 kmol of N2 at 400 kPa and the other compartment contains 7 kmol of CO2 at 200 kPa. Now the partition is...
-
As a precaution, the levers on gasoline pumps in many provinces in Canada must be manually operated at all times. One of the reasons for this is especially relevant in the winter time because the...
-
7. A transformer core is shown in cartoon form in Figure 8.26. This is made up of, perhaps implausibly, 29 gage M-36 laminations: the same sort of material described by Figures 8.19 and 8.20. Assume...
-
2. For a lamp that has a total luminous flux, of 4.5104 lm and total radiant flux, of 300 W, (a) what is the overall luminous efficiency, K for this lamp? (b) if this lamp has a luminous intensity of...

Study smarter with the SolutionInn App


