Reconsider Prob. 1439E. Determine the adiabatic saturation temperature of the humid air. Data From Prob. 1439E: Atmospheric
Question:
Reconsider Prob. 14–39E. Determine the adiabatic saturation temperature of the humid air.
Data From Prob. 14–39E:
Atmospheric air at a pressure of 1 atm and dry-bulb temperature of 90°F has a wet-bulb temperature of 85°F. Using the psychrometric chart.
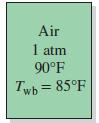
Transcribed Image Text:
Air 1 atm 90°F Twb = 85°F
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 54% (11 reviews)
The pressure temperature and wetbulb temperature of air are specified The adiabatic ...View the full answer

Answered By

Shivani Dubey
Hello guys, I am very good with the Advance mathematics, Calculus, Number theory, Algebra, Linear algebra, statistics and almost all topics of mathematics. I used to give individual tutoring students in offline mode but now want to help students worldwide so I am joining here. I can also give solutions in various coding languages of mathematics and software like mathematica, R, Latex, Matlab, Statistica, etc.
feel free to ask any doubt regarding mathematics
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Thermodynamics An Engineering Approach
ISBN: 9781259822674
9th Edition
Authors: Yunus Cengel, Michael Boles, Mehmet Kanoglu
Question Posted:
Students also viewed these Engineering questions
-
Determine the adiabatic saturation temperature of the humid air in Prob. 14-41. Prob. 14-41. Atmospheric air at a pressure of 1 atm and drybulb temperature of 28oC has a wet-bulb temperature of 20oC....
-
Determine the adiabatic saturation temperature of the humid air in Prob. 14-43E. Prob. 14-43E Atmospheric air at a pressure of 1 atm and drybulb temperature of 90oF has a dew-point temperature of...
-
Reconsider Prob. 1443. Determine the adiabatic saturation temperature of the humid air. Data From Prob. 1443. Atmospheric air at a pressure of 1 atm and dry-bulb temperature of 28C has a dew-point...
-
Dream House Builders, Inc. applies overhead by linking it to direct labor. At the start of the current period, management predicts total direct labor costs of $100,000 and total overhead costs of...
-
One common way to test for authenticity of data is to analyze the frequencies of digits. When people are weighed and their weights are rounded to the nearest pound, we expect the last digits 0, 1, 2,...
-
A social media marketing company is conducting consumer research to see how the income level and age might correspond to whether or not consumers respond positively to a social media campaign. Aliyah...
-
Take the product of the perturbation velocity equation given by Eq. (17.53) by any component of the perturbation velocity. This results in an equation for \(v_{x}^{\prime} v_{y}^{\prime}\), which is...
-
Casey Fisher and Logan Baylor formed a partnership in which the partnership agreement provided for salary allowances of $40,000 and $35,000, respectively. Determine the division of a $20,000 net loss...
-
Please show me the Calculation step by step. Thank you Financial Analysis : Facts: $1 US = $1.40 CDN One truckload / container = 1940 cases One truckload / container = 25 standard pallets One pallet...
-
In free space, E 20 cos (wt 50x) a V/m. Calculate (a) J (b) H (c) w
-
What does a modern air-conditioning system do besides heating or cooling the air?
-
Atmospheric air at a pressure of 1 atm and dry-bulb temperature of 90F has a wet-bulb temperature of 85F. Using the psychrometric chart, determine (a) The relative humidity (b) The humidity ratio (c)...
-
1. The purpose of antitrust policy is to promote _______, which leads to lower _______. 2. There are three types of antitrust policies: (1) _______, (2) _______, and (3) _______. 3. In the Staples...
-
Explain how changing economic and competitive pressure have had an impact on the organization in which you are working or one in which you have worked. How has your business responded to this...
-
Describe the organizational structure of the Microsoft company . Do you think it is optimally organized to achieve its goals? Why or why not?
-
Select 3 different Warehouse Coordinator Job Position for multinational company specializing in Global Sourcing from online jobsites (Position need to be completely presented as advertised with...
-
Gross Income Inclusions Hannah and Josiah, your long time clients are filing a married filing joint tax return. Hannah earned $44,000 during the year, however, due to budget cuts, she was laid off...
-
Define what an AHROP is and how it benefits an organization
-
Joseph Jernigan died in 2013 with a taxable estate of $4.1 million. He was survived by his spouse Josephine and several children. He made taxable gifts of $100,000 in 1974 and $650,000 in 2000. The...
-
The domain of the variable in the expression x 3/x + 4 is________.
-
A process requires a mixture that is 21 percent oxygen, 78 percent nitrogen, and 1 percent argon by volume. All three gases are supplied from separate tanks to an adiabatic, constant-pressure mixing...
-
A mixture of helium and nitrogen with a nitrogen mass fraction of 35 percent is contained in a piston-cylinder device arranged to maintain a fixed pressure of 100 psia. Determine the work produced,...
-
A mixture that is 20 percent carbon dioxide, 10 percent oxygen, and 70 percent nitrogen by volume undergoes a process from 300 K and 100 kPa to 500 K and 400 kPa. Determine the makeup of the mixture...
-
Scenario: Yarning Circle and Talking Stick Sarah has recently been employed by Blue Bay Early Learning Centre. Sarah is very proud of having just achieved the Diploma of Children's Services and she...
-
The principal at your school decides to retire, and several well-qualified teachers on the staff apply for the position. Mr. England has been teaching for four years and has his administrator's...
-
Sandi's parents have been divorced since she was five years old. She is now seven. She lives with her mother and visits her father every other weekend. He brings her to school on Monday mornings...

Study smarter with the SolutionInn App


