The equation of state of a gas is given by where a and b are constants. Use
Question:
The equation of state of a gas is given by
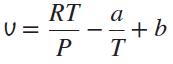
where a and b are constants. Use this equation of state to derive an equation for the Joule-Thomson coefficient inversion line.
Transcribed Image Text:
RT U = a + b T %3D
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
The equation of state of a gas is given by An equation for the JouleThomson coefficient inv...View the full answer

Answered By

Somshukla Chakraborty
I have a teaching experience of more than 4 years by now in diverse subjects like History,Geography,Political Science,Sociology,Business Enterprise,Economics,Environmental Management etc.I teach students from classes 9-12 and undergraduate students.I boards I handle are IB,IGCSE, state boards,ICSE, CBSE.I am passionate about teaching.Full satisfaction of the students is my main goal.
I have completed my graduation and master's in history from Jadavpur University Kolkata,India in 2012 and I have completed my B.Ed from the same University in 2013. I have taught in a reputed school of Kolkata (subjects-History,Geography,Civics,Political Science) from 2014-2016.I worked as a guest lecturer of history in a college of Kolkata for 2 years teaching students of 1st ,2nd and 3rd year. I taught Ancient and Modern Indian history there.I have taught in another school in Mohali,Punjab teaching students from classes 9-12.Presently I am working as an online tutor with concept tutors,Bangalore,India(Carve Niche Pvt.Ltd.) for the last 1year and also have been appointed as an online history tutor by Course Hero(California,U.S) and Vidyalai.com(Chennai,India).
4.00+
2+ Reviews
10+ Question Solved
Related Book For 

Thermodynamics An Engineering Approach
ISBN: 9781259822674
9th Edition
Authors: Yunus Cengel, Michael Boles, Mehmet Kanoglu
Question Posted:
Students also viewed these Engineering questions
-
The van der Waals equation of state, an approximate representation of the behavior of gases at high pressure, is given by Where a and b are constants having different values for different gases. (In...
-
The equation of state of a gas is given as (P + 10/ 2) = RuT, where the units of and P are m3/kmol and kPa, respectively. Now 0.5 kmol of this gas is expanded in a quasi-equilibrium manner from 2 to...
-
The equation of state of a gas is given as v-(P + 10/ v-2) = 5 RuT, where the units of v- and P are m3/kmol and kPa, respectively. Now 0.2 kmol of this gas is expanded in a quasi equilibrium manner...
-
What must be the beta of a portfolio with E( rP ) = 18%, if rf = 6% and E (rM) = 14%?
-
Find the regression equation; unless the problem suggests otherwise, let the first variable be the independent (x) variable. Caution: When finding predicted values, be sure to follow the prediction...
-
Using the employer payroll entry from P7-4B, post the employers share of payroll taxes for the March 11 pay period at Appalachian Limited Home Design to the appropriate General Ledger accounts....
-
Use Table 1, or software, to find (a) \(B(7 ; 18,0.45)\); (b) \(b(7 ; 18,0.45)\); (c) \(B(8 ; 11,0.95)\); (d) \(b(8 ; 11,0.95)\); (e) \(\sum_{k=4}^{11} b(k ; 11,0.35)\); (f) \(\sum_{k=2}^{4} b(k ;...
-
A He-Xe mixture containing 0.75 mole fraction of helium is used for cooling of electronics in an avionics application. At a temperature of 300 K and atmospheric pressure, calculate the mass fraction...
-
1. "Businesses are feeling the impact of the war in Ukraine. Risk managers update their risk profiles due to supply chain disruptions, economic sanctions, and even displaced workforce. These issues...
-
Richard chooses technique 0 and 2 requiring 10+10-20 efforts and provising 10+11=21 benefits. Hence, 21 is returned as the output Example 2: input1: 3 input2: (10,10,10,10) input3: (10,11,12,15)...
-
What is the most general equation of state for which the Joule-Thomson coefficient is always zero?
-
What is the enthalpy departure?
-
Use the GaussJordan elimination method to find all solutions of the system of linear equations. 2y + z- y + +z+ z + -x-9yz + x + y + z x = y - w = 1 w = 14 4w: 11 9 =
-
Write program in Java. You work for a package shipping company and are writing a program to determine shipping rates. The rates are determined by the following rules: If a package weighs less than 16...
-
12. Write Python code that takes a number in base 2 as a string and converts it into a number in base 10. The name of the function is convert. Here is the output: 1 def convert(xstring): 2 #TO DO:...
-
Why is it not possible to have 100% confidence? Explain. (Choose Best Answer Below) a.)A 100% confidence interval is not possible unless either the entire population is sampled or an absurdly wide...
-
Yield to maturity (%) 5.25 5.50 6.00 The following table provides details on three bonds that each have a par value of $100. Assuming annual coupons, what is the forward rate between years 2 and 3?...
-
A fundamental to OS design, is concurrency. What is Concurrency? What are the three contexts causes Concurrency? QUESTION 2: What are the Principles of Concurrency in OS ? QUESTION 3: What is...
-
What would you consider to be the strategy of The Coca-Cola Company: cost leadership or differentiation? Why?
-
As economic conditions change, how do banks adjust their asset portfolio?
-
Why is the combined gas-steam cycle more efficient than either of the cycles operated alone?
-
The gas-turbine portion of a combined gas-steam power plant has a pressure ratio of 16. Air enters the compressor at 300 K at a rate of 14 kg/s and is heated to 1500 K in the combustion chamber. The...
-
Consider a combined gas-steam power plant that has a net power output of 450 MW. The pressure ratio of the gas-turbine cycle is 14. Air enters the compressor at 300 K and the turbine at 1400 K. The...
-
Rewrite the Roman numeral using the least number of symbols possible and the subtractive principle where applicable?
-
Determine, with justification, the smallest possible average of a set whose elements are distinct positive integers that includes the integer 200.
-
This study is for students joining University who are not living in the region. Suppose that a student will join the University next semester and wants to choose between renting a one-bedroom...

Study smarter with the SolutionInn App


