Using Daltons law, show that for a real-gas mixture of k gases, where Z is the compressibility
Question:
Using Dalton’s law, show that
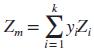
for a real-gas mixture of k gases, where Z is the compressibility factor.
Transcribed Image Text:
Z„ = EyZi %3D i=1
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
Using Daltons law it is to be shown that for a realgas mixture Analysis U...View the full answer

Answered By

BETHUEL RUTTO
Hi! I am a Journalism and Mass Communication graduate; I have written many academic essays, including argumentative essays, research papers, and literary analysis. I have also proofread and written reviews, summaries and analyses on already finished works. I am eager to continue writing!
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Thermodynamics An Engineering Approach
ISBN: 9781259822674
9th Edition
Authors: Yunus Cengel, Michael Boles, Mehmet Kanoglu
Question Posted:
Students also viewed these Engineering questions
-
A gas turbine is operated with gases (Cp = 0.992 kJ/kg K and = 1.29) entering it at 10 bar and 1025C, and leaving it at 1 bar and 550C. Assuming adiabatic flow through the turbine, calculate the...
-
A gas mixture at 300 K and 200 kPa consists of 1 kg of CO2 and 3 kg of CH4. Determine the partial pressure of each gas and the apparent molar mass of the gas mixture.
-
A mixture of Ne and Ar gases at 350 K contains twice as many moles of Ne as of Ar and has a total mass of 50.0 g. If the density of the mixture is 4.00 g/L, what is the partial pressure of Ne?
-
Lucy, a 57-year-old widow, lost her husband in 2021. He was an active participant in the Federal Employees' Retirement System (FERS), and his FERS contributions totaled $27,000 at the time of his...
-
In the previous question, 1350 is an outlier. If we remove this value, show why the sales without outliers are normally distributed at a 0.05 level of significance.
-
The accompanying data file contains 35 observations for an ice cream truck drivers daily income (Income in $), number of hours on the road (Hours), whether it was a particularly hot day (Hot = 1 if...
-
We generated Figure 2.16 by first computing the mean values for each column and then pivoting the values into a single column of mean values. Replicate the data generation steps and use the...
-
The following tasks are completed by the auditor while observing the physical inventory. For each task, state which assertion(s) is tested by the task: (1) existence or occurrence, (2) completeness,...
-
An aluminum wheel rubs against an aluminum plate in such a manner that there is a 51.0 N force of kinetic friction between them, and the relative speed at which the two surfaces slide over each other...
-
Draft the following sentences in active voice. The defendant was attacked by the plaintiff at the beginning of the argument. It is a requirement of good writing skills that active voice be used....
-
A mixture of gases consists of 1 kmol of carbon dioxide, 1 kmol of nitrogen, and 0.3 kmol of oxygen. Determine the total amount of work required to compress this mixture isothermally from 10 kPa and...
-
A 60-L rigid tank contains an ideal-gas mixture of 5 g of N 2 and 5 g of CO 2 at a specified pressure and temperature. If N 2 were separated from the mixture and stored at mixture temperature and...
-
What is the E value for the half-cell on the left-hand side of Figure 20.14? 1.52 V voltmeter - H,(g). 1 atmosphere platinum salt bridge - platinum 298 K H*, 1.00 mol dm-3 MnO, 1.00 mol dm Mn2",...
-
How do ethical considerations factor into decision-making processes within complex business environments ?
-
Bus trip times between 2 stops on a certain route are normally distributed with a mean of 4.7 minutes and a standard deviation of 1.2 minutes. What is the trip time that constitutes the 65th...
-
What role does organizational culture play in mitigating workplace stress and promoting employee well-being?
-
Find f(x + h) - f(x) h f(x)=x-11 for the given function.
-
If X1, X2,...,X, constitute a random sample from an infinite population with the mean and variance a, then find a. the expected value of sample mean X. b. the variance of sample mean X.
-
Carla plans to transfer to a new trust oil and gas properties producing royalty income. She will transfer no other properties. The sole income beneficiary of the trust will be Carla's son, Marshall,...
-
What did Lennox gain by integrating their WMS, TMS, and labor management systems?
-
A reversible absorption refrigerator consists of a reversible heat engine and a reversible refrigerator. The system removes heat from a cooled space at - 15oC at a rate of 70 kW. The refrigerator...
-
An ammonia-water absorption refrigeration cycle is used to keep a space at 25oF when the ambient temperature is 70oF. Pure ammonia enters the condenser at 300 psia and 140oF at a rate of 0.04lbm/s....
-
Describe the Seebeck and the Peltier effects.
-
Choose from the following topics to create discussion: Humanity and the Natural Environment Housing and Home Furnishings Privacy and Socializing
-
On December 31, 2020, Pearl Company had $1,101,000 of short-term debt in the for January 21, 2021, the company issued 22,800 shares of its common stock for $42 per brokerage fees and other costs of...
-
During your interview process for the position of program manager for Booker, Tee, and How you are asked to explain the relationship between cost, schedule, and performance? what is your answer to...

Study smarter with the SolutionInn App


