Tell whether each of the following reactions is likely to be SN1, SN2, E1, E1cB, orE2: NaN3
Question:
Tell whether each of the following reactions is likely to be SN1, SN2, E1, E1cB, orE2:
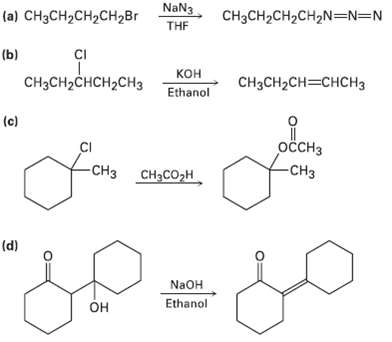
Transcribed Image Text:
NaN3 (a) CHзCH2CH2сH2Br CH3CH2CH2CH,N=N=N THE CI (b) ононалонен, кон CнзCH-CHCH2CHЗ CHзCH2CH—CHCH3 Ethanol (c) оссHз CI -CHз сн-со-н -CHз (d) NaOH Ethanol Он
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 81% (11 reviews)
a CH3CHCHCHBr NaN3 primary The reaction occurs by an SN2 mechani...View the full answer

Answered By

Muhammad adeel
I am a professional Process/Mechanical engineer having a vast 7 years experience in process industry as well as in academic studies as a instructor. Also equipped with Nebosh IGC and lead auditor (certified).
Having worked at top notch engineering firms, i possess abilities such as designing process equipment, maintaining data sheets, working on projects, technical biddings, designing PFD and PID's etc.
Having worked as an instructor in different engineering institutes and have been involved in different engineering resrearch projects such as refinery equipment designing, thermodynamics, fluid dynamics, chemistry, rotary equipment etc
I can assure a good job within your budget and time deadline
4.90+
52+ Reviews
60+ Question Solved
Related Book For 

Question Posted:
Related Video
In this video, A mixture of methanol and air in a large polycarbonate bottle is ignited. The resulting rapid combustion reaction, often accompanied by a dramatic ‘whoosh’ sound and flames, demonstrates the large amount of chemical energy released in the combustion of alcohol
Students also viewed these Organic Chemistry questions
-
Tell whether each of the following reactions is an oxidation, a reduction, orneither. (a) NABH4 H20 CH;CH- CH3CH2CH2OH (b) OH 1. BH3 2. NaOH, H202
-
Tell whether each of the following reactions is an oxidation, a reduction, orneither: (a) CH3CH CH3CH2OH (b) HH + NH NCH2CH2CCH3 Br (c) 1. Mg CHCH2CHCH CCH2CH2CH3 2. H20
-
Tell whether each of the following molecules has a meso stereoisomer. (a) (b) CH CHCH CHCH Cl CH, CHCH2CH,CH CI
-
All individuals in Canada have the responsibility to take meaningful actions towards truth and reconciliation. As business professionals, we take it a step further and talk about our responsibility...
-
What some of the benefits that exist for employees who learn English
-
If a vertical cylinder of water (or any other liquid) rotates about its axis, as shown in FIGURE CP8.71, the surface forms a smooth curve. Assuming that the water rotates as a unit (i.e., all the...
-
A stock price \(S\) is governed by the model where the period length is 1 month. Let \(u=\mathrm{E}[w(k)]\) and \(\sigma^{2}=\operatorname{var}[w(k)]\) for all \(k\). Now suppose the basic period...
-
Using the following information for Gold Star National Bank, calculate that banks ratios of Tier 1 capital-to-risk-weighted assets and total-capital-to-risk-weighted assets. Does the bank have...
-
appears below. Service revenue Operating expenses $ 900,000 700,000 200,000 50,000 Income before income taxes $ 150,000 Income tax expense Net income The following balance sheet information also is...
-
During the current year, Yost Company disposed of three different assets. On January 1 of the current year, prior to the disposal of the assets, the accounts reflected the following: The machines...
-
Which isomer would you expect to undergo E2 elimination faster, trans1-bromo-4-tert-butylcyclohexane or cis-1-bromo-4-tert-butylcyclohexane? Draw each molecule in its more stable chair conformation,...
-
Write the product you would expect from reaction of each of the following alkyl halides with (i) Na + ? SCH3 and (ii) Na + ?? OH (yellow-green = Cl): (a) (c) (b)
-
Nalad Corp. provided the following data related to accounting and taxable income: There are no existing temporary differences other than those reflected in these data. There are no permanent...
-
What is the name of the act of Congress in 2002 intended to bring reform to corporate accountability and stewardship in the wake of a number of major corporate scandals?
-
Juan, a natural person, failed to include information on his return disclosing his participation in a listed transaction. Since Juan had substantial losses from his business, there is no...
-
An employee worked 40 hours at $12 per hour and 5 overtime hours at 1.5 times regular pay. Federal income taxes were deducted from the employee's pay at a rate of 20 percent. Social Security and...
-
When does a while loop invariant need to hold (i.e be true)? (a) O When a program enters the loop. (b) O When a program enters and exits the loop. (c) In the post-condition of the method (d) O At the...
-
QUESTION 31 What contemporary programming languages do not include a goto statement? C# Python C Ruby Java QUESTION 32 The two (2) methods to implement dynamic scoping are? shallow access deep access...
-
Prove Theorem 2.4. That is, prove that \[P\left(\bigcup_{i=1}^{n} A_{i}ight) \leq \sum_{i=1}^{n} P\left(A_{i}ight)\] The most direct approach is based on mathematical induction using the general...
-
The MIT Sloan School of Management is one of the leading business schools in the U.S. The following table contains the tuition data for the masters program in the Sloan School of Management. a. Use...
-
Consider the reaction: A reaction mixture initially contains a CO partial pressure of 1344 torr and a H 2 O partial pressure of 1766 torr at 2000 K. Calculate the equilibrium partial pressures of...
-
Arrange each group of compounds in order of increasing acidity. (a) Phenol, ethanol, acetic acid (b) P-toluenesulfonic acid, acetic acid, chloroacetic acid (c) Benzoic acid, o-nitrobenzoic acid,...
-
What do the following pKa values tell you about the electron-withdrawing abilities of nitro, cyano, chloro, and hydroxyl groups? CH COOH CH,COOH CH COOH CHACOOH CHACOOH NO, 1.68 CN 2.46 CI 2.86 3.83...
-
Given the structure of ascorbic acid (vitamin C): (a) Is ascorbic acid a carboxylic acid? (b) Compare the acid strength of ascorbic acid (pKa = 4.71) with acetic acid. (c) Predict which proton in...
-
Aaron, Deanne, and Keon formed the Blue Bell General Partnership at the beginning of the current year. Aaron and Deanne each contributed $138,000, and Keon transferred an acre of undeveloped land to...
-
The following information pertains to the inventory of Parvin Company: Jan. 1 Apr. 1 Oct. 1 Beginning inventory Purchased Purchased 400 units @ 2,400 units @ 1,100 units $17 $22 $23 During the year,...
-
Gold Nest Company of Guandong, China, makes birdcages for the South China market. The company sells its birdcages through an extensive network of street vendors who receive commissions on their...

Study smarter with the SolutionInn App


