The 1H-NMR spectrum of compound H is as follows, the peaks at highest m/z in the mass
Question:
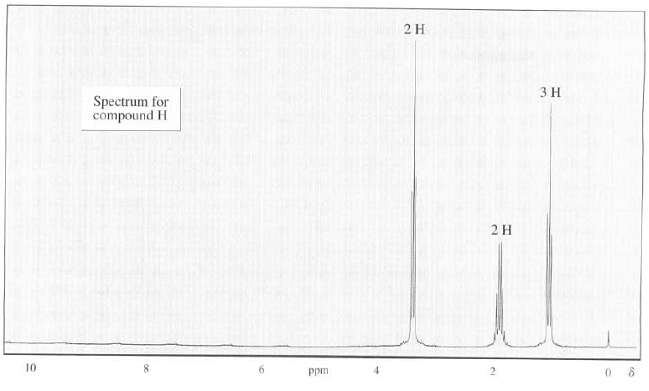
The 1H-NMR spectrum of compound H is as follows, the peaks at highest m/z in the mass spectrum of H appear at 122 and 124 with intensities in a ration of about 1 to 1. Show the structure ofH.
Transcribed Image Text:
2 H ЗН Spectrum for compound H 10 Ppm
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (15 reviews)
The peaks of equal intensity at mz 122 and 124 in the mass spectrum in...View the full answer

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The 1H NMR spectrum of compound A (C8H8O) consists of two singlets of equal area at 5.1 (sharp) and 7.2 ppm (broad). On treatment with excess hydrogen bromide, compound A is converted to a single...
-
The 1H NMR spectrum of the product obtained when an unknown alkene reacts with ozone and the ozonolysis product is worked up under oxidizing conditions is shown. Identify the alkene. 10 (ppm)...
-
The 1H NMR spectrum of N, N-dimethylformamide shows a separate signal for each of the two methyl groups. Can you explain why?
-
Find the antiderivative for each function when C equals 0. a. f(x) = 1 7 b. g(x)== 11 +/- 150 is a. The antiderivative of 5 c. h(x)=4 -- X Part 1 of 3
-
Imagine you are the Director of Internal Audit and the Chairman of the Audit Committee and asks you to prepare a report for the Board of Directors explaining the 2012 COSO framework for internal...
-
Suggest several measures Datotel could use to evaluate the success of its employee retention efforts. Be sure these are practical for a company of a few dozens employees.
-
Calculate the range for each of the following sets of data: a. \(4,6,9\) b. \(14,17,11,19,12\) c. \(25,22,27,30,21,26,29\) d. \(10,8,4,16,9,7,9,13,6,11\) e. \(73,66,91,84,69,87,62,79,82,90\) f....
-
On November 1, 2017, Nordin Inc. (Nordin) issued a $5,000,000 bond with a 6 percent coupon rate and a maturity date of October 31, 2022. Interest is paid semi-annually on April 30 and October 31. The...
-
An eight year note for $3700 issued on November 1, 2015, with interest at 5.8% compounded annually is discounted on September 1, 2016, at 8.9% compounded semi-annually. What are the proceeds of the...
-
A body of mass m is thrown straight up with velocity vo. Find the velocity v' with which the body comes down if the air drag equals kv2, where k is a constant and v is the velocity of the body.
-
Compounds E and F are isomers with the formula C6H8 both react with H2 in the presence of Pt to give G (C6H12), G shows a single peak in its 13C-NMR spectrum. E has no absorption maximum above 200nm...
-
Ultraviolet spectroscopy of often used to monitor the amount of a protein in a sample, the amount of protein is correlated with the absorbance at 280nm. Explain which of the following amino acids you...
-
The stockholders equity section of Plaka Corporations balance sheet follows. Assuming one years dividend in arrears, determine the book value per share for both the preferred and the common stock....
-
Consider the following class. public class Person (private String name; public Person(String name) { this.name = } name; @Override public String tostring() { return this.getClass().getName() +...
-
You have been provided with the following information for the city of Athena: General Fund: Cash: $500,000 . . Property Tax Receivable: $200,000 Accounts Payable: $100,000 Fund Balance: $600,000 Land...
-
Corporation is a company which handles the auction of the different antiques. The company wants to shift from the offline system to the online auction system. For this they have consulted a database...
-
large sampling multi-volume encyclopedia like the Encyclopaedia Britannica. Describe in detail the ways in which you would organise and index the encyclopedia text, and the facilities you would offer...
-
What are the probabilities of outcomes 0 and 1 if the first qubit of |i is measured? (ii) What are the probabilities of outcomes 0 and 1 if the second qubit of |i is measured? (iii) What is the state...
-
Todd is working on resource scheduling in preparation for the start of a project. There is a potential problem in the works, however, as the new collective bargaining agreement with the companys...
-
The relationship described in question 7 does not always appear to hold. What factors, besides the number of firms in the market, might affect margins?
-
Every year since 1995, there has been a Fed Challenge. This is a contest sponsored by the Federal Reserve Board and Citibank. More than 200 high school students throughout the country participate....
-
Complete the following acid-base reactions: (a) HCCH + NaH
-
What is the conjugate base of each of the following acids? (a) NH3 (b) H2O (c) H2 (d) HCiCH (e) CH3OH (f) H3O+
-
Write equations showing the Lewis acid-base reaction that takes place when: (a) Methanol (CH3OH) reacts with BF3. (b) Chloromethane (CH3Cl) reacts with AlCl3. (c) Dimethyl ether (CH3OCH3) reacts with...
-
You have $8,500 to invest for 5 years. Find how much you'll have at the end of the 5 years if you earn 3.4% interest compounded (a) annually: $ (b) monthly: $ (c) daily: $ (d) continuously: $
-
Create instructions for a UHD student on how to use Zoom to record an oral presentation. This assignment has two purposes: I need to ensure you know how to use Zoom to record your group's oral...
-
Solve for x: 1 3x-3x-2=24 2 3*+1=3x+1/3 3 3x+3x+2=10 4 5x+1 1 =2.5* 5 2x+2x+2x=96 6 12x+12=288 7 2x+2x-2=5 8 22x+4x+1=80 EXERCISE 5 Solve for x: 8.4141-32 = 1 2 3x+1=9x. 27 3 3x+2 = 9.27 4 5-1. = 25*...

Study smarter with the SolutionInn App


