The Favorskii reaction involves treatment of an ?-bromo ketone with base to yield a ring-contracted product. For
Question:
The Favorskii reaction involves treatment of an ?-bromo ketone with base to yield a ring-contracted product. For example, reaction of 2-bromocyclo-hexanone with aqueous NaOH yields Cyclopentanecarboxylic acid. Propose a mechanism.
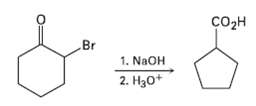
Transcribed Image Text:
сoон Br 1. NAOH 2. Нао*
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 76% (13 reviews)
H Br loss of proton at a posit...View the full answer

Answered By

GERALD KAMAU
non-plagiarism work, timely work and A++ work
4.40+
6+ Reviews
11+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Propose a mechanism for the reaction of cyclohexyl methyl ketone with excess bromine in the presence of sodium hydroxide.
-
Propose a mechanism for the reaction of benzoic acid with oxalyl chloride. This mechanism begins like the thionyl chloride reaction, to give a reactive mixed anhydride. Nucleophilic acyl substitution...
-
Reaction of benzanilide with chlorine in acetic acid yields a mixture of two monochloro derivatives formed by electrophilic aromatic substitution. Suggest reasonable structures for these two isomers....
-
Jinny Buffett recently retired as a flight attendant and is interested in opening a fitness center and health spa exclusively for women in Grand Cayman, where she resides. After careful study, she is...
-
Which silent messages does a business letter convey?
-
Read the profile of Harry Rosen. Identify why Harry has been so successful in marketing to the high-end menswear market segment.
-
Give examples of cases where work is done on a system without changing its kinetic energy.
-
Rocky Mountain Interiors deposits all cash receipts each Wednesday and Friday in a night depository, after banking hours. The data required to reconcile the bank statement as of July 31 have been...
-
In a high-profile criminal trial, the defendant alleges that their confession was coerced by law enforcement officers during interrogation. How might the court assess the admissibility of the...
-
This exercise refers to the drunk driving panel data regression summarized in Table 10.1. a. New Jersey has a population of 8.1 million people. Suppose that New Jersey increased the tax on a case of...
-
One of the later steps in glucose biosynthesis is the isomerization of fructose 6-phosphate to glucose 6-phosphate. Propose a mechanism, using acid or base catalysis asneeded. C- - - C=0 - - - - - -...
-
Treatment of a cyclic ketone with diazomethane is a method for accomplishing a ring-expansion reaction. For example, treatment of Cyclohexanone with diazomethane yields Cycloheptanone. Propose...
-
What is the parallax angle for a star that is 55ly away? How many parsecs is this?
-
Use Transition and Markov to determine the page ranks for the eightpage graph shown below. Eight-page example (6)
-
Cover time. Write a program that estimates the time required for the random surfer to visit every page at least once, starting from a random page.
-
Write a static method multiply() that takes two square matrices of the same dimension as arguments and produces their product (another square matrix of that same dimension). Extra credit : Make your...
-
Suppose that your page is page 23 in medium.txt. Is there a link that you could add from your page to some other page that would lower the rank of that page? public class IntOps { } public static...
-
What are the main aspects of changeability?
-
What is the difference between relationship-based governance and rules-based governance? Why is it important to the development of capital markets?
-
Find the radius of convergence in two ways: (a) Directly by the CauchyHadamard formula in Sec. 15.2. (b) From a series of simpler terms by using Theorem 3 or Theorem 4.
-
According to VSEPR theory, what determines the geometry of a molecule?
-
Give the product expected when benzoic acid reacts with each of the following reagents. (a) CH3I, K2CO3 (b) Concentrated HNO3, H2SO4 (c) PCl5 (d) P2O5, heat
-
How many milliliters of aqueous 0.1 M NaOH are required to form the disodium salt from 100 mg of succinic acid?
-
Give the product(s) formed and the curved-arrow notation for the reaction of 0.01 mole of each reagent below with 0.01 mole of acetic acid. (a) Cs+ -OH (b) H3C--Li (c) NaH
-
Shawn Inc. proposed to install one Robotic Machine for his new project, an initial investment of CANVAS Technology will be $520,000 and the Robotic will be expected to generate net cash flows of at...
-
You anticipate the receipt of money in 200 days, which you will use to purchase stocks in a particular company. The stock is currently selling for $51 and will pay a $0.5 dividend in 50 days and...
-
1) Based on the stock chart for Michaels Companies Inc, what do you think the short and long-term growth potentials are for this company? (discuss the advantages/disadvantages) Link to the stock...

Study smarter with the SolutionInn App


