The figure shows spectra of 1.0010 -4 M MnO - 4 , 1.0010 -4 M Cr 2
Question:
The figure shows spectra of 1.00×10-4 M MnO-4, 1.00×10-4 M Cr2O72-, and an unknown mixture of both, all in 1.000-cm-pathlength cells. Absorbances are given in the table. Use the least-squares procedure in Figure 18-3 to find the concentration of each species in the mixture.
-1.png)
-2.png)
Figure 18-3
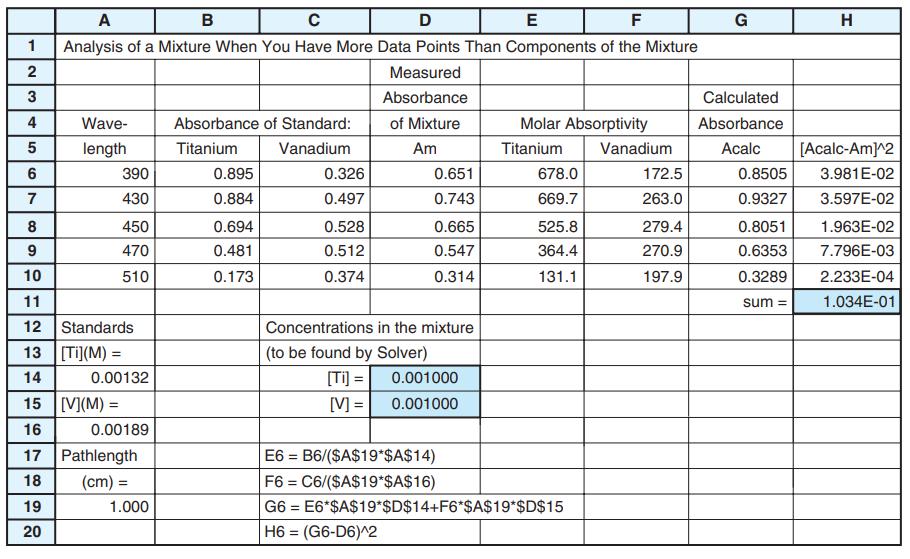
Transcribed Image Text:
Wavelength (nm) MnO standard Cr andard Mixture 266 288 320 350 360 0.042 0.082 0.168 0.125 0.056 0.410 0.283 0.158 0.318 0.181 0.766 0.571 0.422 0.672 0.366 0.9 0.8 Mixture 8 0.6 0.5 0.4 0.3 Mno 0.1 250 300 350 400 450 500 550 600 650 Wavelength (nm)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 28% (7 reviews)
1 2 3 4 Wave 5 length 56 7 8 69 A B D E F G Analysis of a Mixture When ...View the full answer

Answered By

Mamba Dedan
I am a computer scientist specializing in database management, OS, networking, and software development. I have a knack for database work, Operating systems, networking, and programming, I can give you the best solution on this without any hesitation. I have a knack in software development with key skills in UML diagrams, storyboarding, code development, software testing and implementation on several platforms.
4.90+
56+ Reviews
137+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Chemical Engineering questions
-
Consider the reaction 3A + B + C D + E where the rate law is defined as An experiment is carried out where [B]0 = [C]0 = 1.00 M and [A]0 = 1.00 Ã 10-4 M. a. If after 3.00 minutes [A] = 3.26...
-
Gold nanoparticles (Figure 16-29) can be titrated with the oxidizing agent TCNQ in the presence of excess of Br to oxidize Au(0) to AuBr in deaerated toluene. Gold atoms in the interior of the...
-
The figure shows a rear view of a loaded two-wheeled wheelbarrow on a horizontal surface. It has balloon tires and a weight W = 684 N, which is uniformly distributed. The left tire has a contact area...
-
Glass bottles can be either recycled (crushed and re-melted) or reused. The market will tend to choose the cheapest path. What factors will tend to affect the relative cost of these options? Is the...
-
At December 31, 2014, the trial balance of Markowitz Company contained the following amounts before adjustment. Instructions (a) Prepare the adjusting entry at December 31, 2014, to record bad debt...
-
The payroll register of Seaside Architecture Company indicates $870 of Social Security and $217 of Medicare tax withheld on total salaries of $14,500 for the period. Assume earnings subject to state...
-
A smart contract is a computerized transaction protocol that executes the terms of a contract. The general objectives are to satisfy common contractual conditions (such as payment terms, liens,...
-
A researcher uses multiple regression to predict a clients sales volume based on gross domestic product, personal income, disposable personal income, unemployment, and the consumer price index. What...
-
which a disk A of radius r rotates about a fixed vertical axis with angular displacement q1. A bar B of length h is pinned to A such that the axis of rotation is a radial line of A. (Note this is a...
-
The National Weather Service issues precipitation forecasts that indicate the likelihood of measurable precipitation ( .01 inch) at a specific point (the official rain gauge) during a given time...
-
Explain how signal amplification is achieved in enzymelinked immunosorbent assays.
-
What is the advantage of a time-resolved emission measurement with Eu 3+ versus measurement of fluorescence from organic chromophores?
-
Write 84.175, -528.685, 0.000924138, and -362005 in floating-point form, rounded to 5S (5 significant digits).
-
Gate Corp. acquired all of Way Corp's assets in a Type C reorganization on October 19, 2019. On the date of acquisition, Way Corp. had an unused net capital loss of $70,000. Gate Corp. had a net...
-
Are you a morning or an evening person? When did you first decide that you were one or the other? Has the fact that you are a morning, or an evening person affected your career decisions in any way?
-
How many salts are there?
-
How is an acid different from a base?
-
Equipment for manufacturing vegetable oil products is purchased from Alfa. Items such as oil expellers, filter presses, and a steam generator are purchased for $1.2 million. These devices are...
-
Peter Wolfgang is considering the purchase of shares in a closed-end mutual fund. The NAV is equal to 22.50, and the latest close is 20.00. Is this fund trading at a premium or discount? How big is...
-
Planning: Creating an Audience Profile; Collaboration: Team Projects. Compare the Facebook pages of three companies in the same industry. Analyze the content on all available tabs. What can you...
-
Consider the titration of 50.0 mL of 0.050 0 M malonic acid with 0.100 M NaOH. Calculate the pH at each point listed and sketch the titration curve: V b = 0.0, 8.0, 12.5, 19.3, 25.0, 37.5, 50.0, and...
-
Write the chemical reactions (including structures of reactants and products) that occur when the amino acid histidine is titrated with perchloric acid. (Histidine is a molecule with no net charge.)...
-
Select indicators from Table 10-3 that would be useful for the titrations in Figures 10-1 and 10-2 and the pK a = 8 curve in Figure 10-3. Select a different indicator for each titration and state...
-
How crucial is it to understand the many sorts of speech contexts on a daily basis? Give one specific example. Here are the types of speech context: 1. Public Communication 2. Small Group...
-
Enmebaragesi of Kish, Inc. has sales of $282,000, cost of goods sold of $206,000, depreciation of $4,200, and interest expense of $22,000. The tax rate is 30.8 percent. What is the times interest...
-
Inkishush and Company has a book value per share of $10.35, earnings per share of $2.23, and a price-earnings ratio of 19.4. What is the market-to-book ratio?

Study smarter with the SolutionInn App


