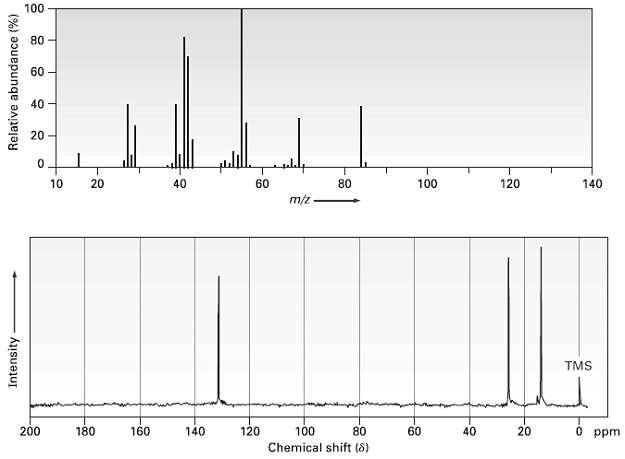
The mass spectrum and 13C NMR spectrum of a hydrocarbon are shown. Propose a structure for this
Question:
The mass spectrum and 13C NMR spectrum of a hydrocarbon are shown. Propose a structure for this hydrocarbon, and explain the spectraldata.
Transcribed Image Text:
100 80 60 40 20 aly 120 10 40 60 80 100 140 m/z TMS 200 180 160 120 100 80 60 20 0 ppm 140 40 Chemical shift (8) Intensity Relative abundance (%) 20
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 46% (13 reviews)
The peak in the mass spectrum at mz 84 is probably the molecular ion of the unknown compound and co...View the full answer

Answered By

Rajat Gupta
used to take tution classes from my school time.
Conducted special topic claases during my graduation to help the students pass their exams.
Currently, teaching and conducting online claases during my post- graduation too.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The proton and 13C NMR spectra of a compound of formula C4H11N are shown here. Determine the structure of this amine, and give peak assignments for all of the protons in the structure. 200 180 160...
-
An unknown, foul-smelling hydrocarbon gives the mass spectrum and infrared spectrum shown. (a) Use the mass spectrum to propose a molecular formula. How many elements of unsaturation are there? (b)...
-
The mass spectrum and infrared spectrum of an unknown compound are shown in Figures 13.27 and 13.28, respectively. Identify the compound. Figure 13.27 The mass spectrum for Problem 28. 100 E 80 3 60...
-
The Holtz Corporation acquired 80 percent of the 100,000 outstanding voting shares of Devine, Inc., for $7.20 per share on January 1, 2014. The remaining 20 percent of Devines shares also traded...
-
It is estimated that a firm contemplating entering the breakfast cereal market would need to invest $100 million to build a minimum efficient scale production plant (or about $10 million annually on...
-
Why is the purchase of rental real estate often referred to as a tax-sheltered investment?
-
Seaforth Sunglasses had the following transactions in December. The beginning inventory on 1 December consisted of 320 pairs of sunglasses at $80 each. Required (a) Prepare journal entries to record...
-
Refer to the information reported about Gazelle Corporation in Problem 16- 1B. In Problem 16- 1B, Gazelle Corporation, a merchandiser, recently completed its calendar- year 2013 operations. For the...
-
a) i] Define the term management? [1 marks] ii] Managers must have specific skills and play certain roles in organizations if they are to inspire employees to meet organizational objectives; explain...
-
The following are the statements of financial position of two companies at 31 October 2018, the end of their most recent financial years: The following additional information is available: 1). On 1...
-
Propose structures for the three compounds whose 1H NMR spectra are shown. (a) C 5 H 10 O (b) C 7 H 7 Br (c) C 8 H 9 Br TMS O ppm 10 Chemical shift (8) TMS O ppm 10 9. 8. 3 2 Chemical shift (8) TMS O...
-
Compound A, a hydrocarbon with M + = 96 in its mass spectrum, has the 13 C spectral data that follow. On reaction with BH 3 followed by treatment with basic H 2 O 2 , A is converted into B, whose 13...
-
Every day, millions of people use Facebook to connect with friends, relatives, and businesses. As a result, Facebook is now the world's leading social media company. In your opinion, what factors...
-
What elements make an Islamic contract defective or invalid besides the prohibited elements in question 8? Question 8 Which of the following is NOT a condition of the subject matter of the Islamic...
-
Using the information in Question 1, and on the basis of AAOIFIs standard on investments, you are required to provide (for each of the 5 years) the following: a. The accounting entries in the books...
-
Which stage of the decision-making process is most affected by the dimensions of social commerce? Explain.
-
Why are sukuk becoming an increasingly important source of financing for companies? Discuss.
-
If necessary, we will (review, change, reduce) overtime hours to (fix, balance, rework) the budget. From the choices in parentheses, select the most precise, vigorous words.
-
A large-sized chemical company is considering investing in a project that costs `5,00,000. The estimated salvage value is zero; tax rate is 35 per cent. The company uses straight line method of...
-
Federated Shipping, a competing overnight delivery service, informs the customer in Problem 65 that they would ship the 5-pound package for $29.95 and the 20-pound package for $59.20. (A) If...
-
Potassium nitrate has a lattice energy of -163.8 kcal/mol and a heat of hydration of -155.5 kcal/mol. How much potassium nitrate has to dissolve in water to absorb 1.00 * 10 2 kJ of heat?
-
Show how Diels-Alder reactions might be used to synthesize the following compounds. (a) (b) (c) (d) (e) (f) (g) (h) (i) CH 3COOCH CH3 CN CN CI CI Cl Cl Cl CI CI C chlordane CI CI Cl Cl CI C aldrin CN...
-
Furan and maleimide undergo a Diels-Alder reaction at 25 °C to give the endo isomer of the product. When the reaction takes place at 90 °C, however, the major product is the exo isomer....
-
(a) Sketch the pi molecular orbitals of hexa-1, 3, 5-triene (Figure 15-25). (b) Show the electronic configuration of the ground state of hexa-1, 3, 5-triene. (c) Show what product would result from...
-
Microblogging is found in Web 2.0. Explain clearly and in detail why microblogging could only exist in, and be a product of, Web 2.0, and not exist in or be a product of Web 1.0.
-
The brute force algorithm for string matching is given below: ALGORITHM Brute Force String Match(T[0..n-1], P[0..m-1]) //Implements brute-force string matching //Input: An array T[0..n-1] of n...
-
Below is a 2-3 tree, into which 4 elements were inserted so far. Please insert C,H,X,P and L in this tree in the given order. Show all intermediate steps with your drawings. E RS

Study smarter with the SolutionInn App


