The metal ion indicator xylenol orange (Table 11-3) is yellow at pH 6 ( max = 439
Question:
The metal ion indicator xylenol orange (Table 11-3) is yellow at pH 6 (λmax = 439 nm). The spectral changes that occur as VO2+ is added to the indicator at pH 6 are shown here. The mole ratio VO2+/xylenol orange at each point is
-1.png)
Suggest a sequence of chemical reactions to explain the spectral changes, especially the isosbestic points at 457 and 528 nm.
-2.png)
Table 11-3
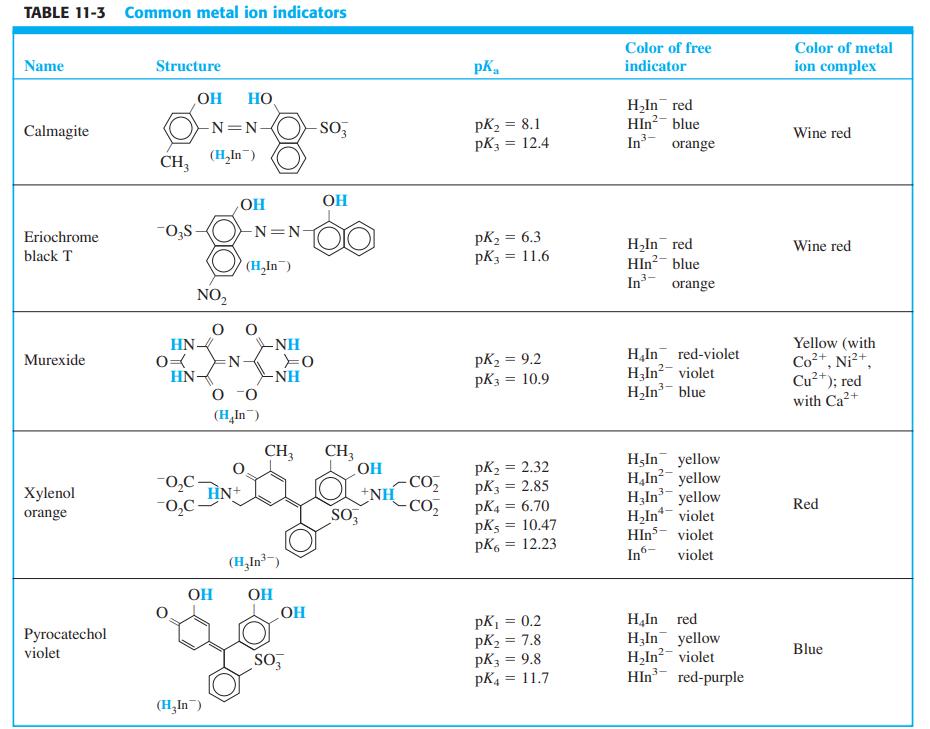
Transcribed Image Text:
Trace Mole ratiTrace Mole ratio Trace Mole ratio 0.60 0.70 0.80 0.90 1.0 12 13 1.5 14 2.0 15 3.1 16 4.1 1.3 0.10 2 0.20 0.30 0.40 5 0.50 4 10 16 0.1 Absorbance unit 13 12 600 500 Wavelength (nm) 400
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 90% (11 reviews)
As VO 2 is added traces 19 the peak at 439 decreases a...View the full answer

Answered By

Nyron Beeput
I am an active educator and professional tutor with substantial experience in Biology and General Science. The past two years I have been tutoring online intensively with high school and college students. I have been teaching for four years and this experience has helped me to hone skills such as patience, dedication and flexibility. I work at the pace of my students and ensure that they understand.
My method of using real life examples that my students can relate to has helped them grasp concepts more readily. I also help students learn how to apply their knowledge and they appreciate that very much.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Chemical Engineering questions
-
A 1.000-mL sample of unknown containing Co2+ and Ni2+ was treated with 25.00 mL of 0.03872 M EDTA. Back titration with 0.02127 M Zn2+ at pH 5 required 23.54 mL to reach the xylenol orange end point....
-
A novel metal ion adsorbent material based on the bio polymer poly glucosamine is cast into a gel bead. The amine groups on the bio polymer have a high affinity for transition metal ions at...
-
The amount of lactic acid, HC3H5O3, produced in a sample of muscle tissue was analyzed by reaction with hydroxide ion. Hydroxide ion was produced in the sample mixture by electrolysis. The cathode...
-
Does the fact that the strategic petroleum reserve has never been used to offset shortfalls caused by an embargo mean that the money spent in creating the reserve has been wasted? Why or why not?
-
Farwell Company closes its books monthly. On September 30, selected ledger account balances are: Notes Receivable .......$37,000 Interest Receivable ....... 183 Notes Receivable include the...
-
1. What should Andy Gibson have done differently in his meeting with Jacob Clemson in Jacobs office? 2. What are some things Andy could have done differently from his initial contact with Mr. Clemson...
-
Given the different needs of the HRP/workforce planning process discussed in this chapter, what types of data would you expect the HRIS data warehouse to contain?
-
Storico Co. just paid a dividend of $3.85 per share. The company will increase its dividend by 20 percent next year and will then reduce its dividend growth rate by 5 percentage points per year until...
-
You are evaluating a project with the following cash flows: initial investment is $ - 1 1 , and the expected cash flows for years 1 - 3 are $ 6 , $ 1 4 and $ 1 1 ( all cash flows are in millions of...
-
Prepare T-account entries for all appropriate (see tips below) B/S and I/S accounts to obtain a 3,000 debit balance in the C&CEMain T-account. Prepare the T-accounts under the Direct method approach....
-
When are isosbestic points observed and why?
-
Infrared spectra are customarily recorded on a transmittance scale so that weak and strong bands can be displayed on the same scale. The region near 2 000 cm -1 in the infrared spectra of compounds A...
-
The following information was obtained for Lisas Fashionista Ltd for the year ended 30 June 2020. Required (a) Prepare a statement of profit or loss and other comprehensive income for Lisas...
-
A Social Security number consists of nine digits in a particular order, and repetition of digits is allowed. If randomly selecting digits for one Social Security number, what is the probability that...
-
Favorite musical group Classify the variable as qualitative or quantitative.
-
Telephone interviews of 1,502 adults found that only 69% could identify the current vice-presidents voice over the phone. Determine whether the underlined value is a parameter or a statistic.
-
Address Classify the variable as qualitative or quantitative.
-
In a survey of a sample of 1050 teenagers, 17% said they like to watch soccer. Determine whether the underlined value is a parameter or a statistic.
-
How are stellar masses determined?
-
Consider a closed, rigid tank with a volume of 0.8L, filled with cold water initially at 27C. The tank is filled such that there are no voids (air pockets) within. The initial pressure within the...
-
Select indicators from Table 10-3 that would be useful for the titrations in Figures 10-1 and 10-2 and the pK a = 8 curve in Figure 10-3. Select a different indicator for each titration and state...
-
When 100.0 mL of a weak acid were titrated with 0.093 81 M NaOH, 27.63 mL were required to reach the equivalence point. The pH at the equivalence point was 10.99. What was the pH when only 19.47 mL...
-
A 0.100 M solution of the weak acid HA was titrated with 0.100 M NaOH. The pH measured when V b = V e was 4.62. Using activity coefficients, calculate pKa. The size of the A - anion is 450 pm.
-
6x+17x-3 Evaluate the following limit: lim xo 5x2+14x-3
-
Find all the critical numbers of y=x-2x-3, then determine the local minimum and maximum points by using a graph.
-
At September 3 0 , the end of Beijing Companys third quarter, the following stockholdersequity accounts are reported. Common stock, $ 1 0 par value $ 4 8 0 , 0 0 0 Paid - in capital in excess of par...

Study smarter with the SolutionInn App


