What carbonyl compounds might you start with to prepare the following compounds by Grignard reaction? List all
Question:
What carbonyl compounds might you start with to prepare the following compounds by Grignard reaction? List all possibilities.
(a) 2-Methyl-2-propanol
(b) 1-Ethylcyclohexanol
(c) 3-Phenyl-3-pentanol
(d)2-Phenyl-2-pentanol
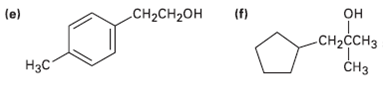
Transcribed Image Text:
(f) CH-CH2он (e) он CH2CCH3 Нэс CHз
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (17 reviews)
Grignard Reagent Carbonyl Compound a b CH3MgBr 2 CHMgBr or CH3CHMg Br i CH3CCH...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
What alkynes would you start with to prepare the followingketones? (a) (b) CHCHH2H CH3CH2CH2CH3
-
What alkyne would you start with to prepare each of the following compounds by a hydroboration/oxidationreaction? (b) (a) CH CH-CCHCH3 -CH2CH CH
-
What ester an what Grignard reagent might you start with to prepare the following alcohols? (b) , H (c) HCH-H2H2,H CH-CH (a)
-
Oliveras Company had net credit sales during the year of $800,000 and cost of goods sold of $500,000. The balance in accounts receivable at the beginning of the year was $100,000, and the end of the...
-
Describe the differences between multinational and global firms.
-
Compare the means of each pair of groups using the Bonferroni method? Pulmonary Disease Twenty-two young asthmatic volunteers were studied to assess the short-term effects of sulfur dioxide (SO 2 )...
-
The probabilities that a TV station will receive \(0,1,2,3, \ldots, 8\) or at least 9 complaints after showing a controversial program are, respectively,...
-
Given the following network, with activity times in months, determine the earliest and latest activity times and slack for each activity. Indicate the critical path and the projectduration. 10 6 12 8...
-
The David Derrick Insurance company wants to sells a certain policy for $1000 to John Graham Jr. They know from historic data that there is a 1 in 100 chance John will file a $20,000 claim, a 1 in...
-
Rachel Sells is unable to reconcile the bank balance at January 31. Rachel's reconciliation is shown here. Cash balance per bank.......................................$3,677.20 Add: NSF...
-
How would you carry out the following transformations? Co .CO2H (a) (b) Co CH2 C (c) CH2SH
-
Evidence for the intermediate carbocations in the acid-catalyzed dehydration of alcohols comes from the observation that rearrangements sometimes occur. Propose a mechanism to account for the...
-
During an interview with a reporter, former Microsoft CEO Steve Ballmer discussed the data compiled on the usafacts.org Web site. Ballmer asked if the reporter knew how many people the government...
-
Penny is 25 and has a dream of accumulating $3,000,000 by the time she is 50 (in 25 years) so that she can retire. She expects to achieve annual returns of 10% on her investment. Assuming she has $0...
-
Family violence causes significant pain to many who encounter the family law system. Discuss the prevalence of family violence in Australia and assess whether the family courts currently consider and...
-
On January 2, 2025, Big Inc. acquired 15% of Small Co.'s outstanding common stock for $62,400 and did not exercise significant influence. Small earned net income of $96,000 in 2025 and paid dividends...
-
You own a shop and you produce widgets that require the use of two raw components: steel and copper. The unit price of a widget is 10 USD. You bought 10,000 Kg of copper at a price per kilo of 9.76...
-
I owe $46k on a $50k business loan that I got and purchased a home with. Tree fell on home got substantial insurance claim. I just sold home home for $85k. I made a $35k profit after closing costs....
-
What court or courts in your state have jurisdiction to hear appeals from felony convictions? What changes, if any, would you propose for your states system of appellate courts?
-
An annual report of The Campbell Soup Company reported on its income statement $2.4 million as equity in earnings of affiliates. Journalize the entry that Campbell would have made to record this...
-
In a 1999 study of cobalt nanocrystals, D. P. Dinega and M. G. Bawendi discovered that cobalt forms an interesting cubic structure unlike any of the cubic structures described in this chapter. They...
-
Fischer projection formulas are often used to depict compounds such as glyceraldehyde, lactic acid, and tartaric acid. Draw Fischer projections for both enantiomers of (a) Glyceraldehyde, (b)...
-
Designate the (R) or (S) configuration at each chirality center in the following molecules. Cl SH Br Br
-
Albuterol, shown here, is a commonly prescribed asthma medication. For either enantiomer of albuterol, draw a three-dimensional formula using dashes and wedges for bonds that are not in the plane of...
-
1 B C Use Excel to journalize the adjusting entries. Use the blue shaded areas for inputs. (Always use cell references and formulas where appropriate to receive full credit. If you copy/paste from...
-
A. Make each pair of radicals similar by reducing the radicand. 1. 2,8 2. 3.24 3. 45,125 4. 3,96 5. 27.75 6. 48,12 7. 28,63 8. 4/32,1/162 9. 40,135 10. 8.200 B. Referring to each pair in letter A, do...
-
Assume that you are the president of Highlight Construction Company. At the end of the first year of operations (December 31), the following financial data for the company are available: Cash...

Study smarter with the SolutionInn App


