A 13C NMR spectrum of commercially available 2, 4-pentanediol, shows five peaks at 23.3, 23.9, 46.5, 64.8,
Question:
A 13C NMR spectrum of commercially available 2, 4-pentanediol, shows five peaks at 23.3, 23.9, 46.5, 64.8, and 68.1 ?. Explain.
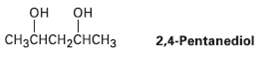
Transcribed Image Text:
он он CHзCнсH-CHCH3 2,4-Pentanediol
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (14 reviews)
Commercial 24pentanediol is a mixture of three stereoisomers RR SS and RS The meso isomer sh...View the full answer

Answered By

Nyron Beeput
I am an active educator and professional tutor with substantial experience in Biology and General Science. The past two years I have been tutoring online intensively with high school and college students. I have been teaching for four years and this experience has helped me to hone skills such as patience, dedication and flexibility. I work at the pace of my students and ensure that they understand.
My method of using real life examples that my students can relate to has helped them grasp concepts more readily. I also help students learn how to apply their knowledge and they appreciate that very much.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The mass spectrum and 13C NMR spectrum of a hydrocarbon are shown. Propose a structure for this hydrocarbon, and explain the spectraldata. 100 80 60 40 20 aly 120 10 40 60 80 100 140 m/z TMS 200 180...
-
A compound C6H14O has the 13C NMR spectrum shown in Figure 15.7. Its mass spectrum has a prominent peak at m/z 31. Suggest a reasonable structure for this compound.
-
A compound (C3H7ClO2) exhibited three peaks in its 13C NMR spectrum at 46.8 (CH2), 63.5 (CH2), and 72.0 ppm (CH). Excluding compounds that have Cl and OH on the same carbon, which are unstable, what...
-
Using the SEDAR database, find the most recent annual reports for two Canadian retailers (e.g., Loblaw, Rona, Danier Leather). Required: a. Based on the information provided in the companies audited...
-
In rapidly developing economies --- such as India and South Korea --- conglomerates are far more common than they are in the US and Western Europe. Use the BCG growth/share matrix to explain why this...
-
At 1000 K, Kp = 1.85 for the reaction (c) What is the value of Kc for the reaction in part (b)?
-
Define concept growth. What does this mean to entrepreneurial firms?
-
Laminar flow in a triangular duct (Figure 3B.2) 2 one type of compact heat exchanger is shown in Figure 3B.2 (a). In order to analyze the performance of such an apparatus, it is necessary to...
-
How does the organization's culture, including its norms, values, and beliefs, impact the different dimensions of organizational commitment ?
-
Let today be November 3, 2008. (a) Use the LIBOR rate and the swap data on November 3, 2008 in Table 11.26 and fit the LIBOR curve. (b) From the LIBOR discount curve, fit the Ho-Lee model of the...
-
3-Methyl-2-butanol has five signals in its 13C NMR spectrum at 17.90, 18.15, 20.00, 35.05, and 72.75 ?. Why are the two methyl groups attached to C3 nonequivalent? Making a molecular model should be...
-
Carboxylic acids (RCO 2 H) react with alcohols (R?OH) in the presence of an acid catalyst. The reaction product of propanoic acid with methanol has the following spectroscopic properties. Propose a...
-
Researchers wanted to compare two drugs, for-moterol and salbutamol, in aerosol solution, for the treatment of patients who suffer from exercise-induced asthma.2 Patients were to take a drug, do some...
-
Tax credits are an important way to offset a taxpayer's total tax liability dollar for dollar. Within the Internal Revenue Code (IRC), many tax credits are included to help reduce taxpayers' taxes...
-
Discuss the advantages and disadvantages of the historical cost presentation under US GAAP compared with the fair market value presentation under IFRS. Explain which approach you believe is better...
-
1. List and describe the three types of manufacturing costs. 2. Describe the differences between product costs and period costs. 3. The management of a manufacturing company always has the...
-
There is Parent company and their Sub Company that was acquired on the 1st of July 2022. Now that the financial year is over we need to prepare the consolidation journal entries, pre-acquisition...
-
Sit 'N Eat family restaurant serves three menus during the day, 7 days a week The restaurant has 25 tables that each seat 4 people The restaurant experiences a seat turnover of 2 for each menu...
-
A janitor is pushing an \(11-\mathrm{kg}\) trashcan across a level floor at constant speed. The coefficient of friction between can and floor is 0. 10 . (a) If the janitor is pushing horizontally,...
-
The nitrogen atoms in N2 participate in multiple bonding, whereas those in hydrazine, N2H4, do not. (a) Draw Lewis structures for both molecules. (b) What is the hybridization of the nitrogen atoms...
-
Which molecule would you expect to be more soluble in water: CCl 4 or CH 2 Cl 2 ?
-
Show which of the nitrogen atoms in purine are basic, and which one is not basic. For the non-basic nitrogen, explain why its nonbonding electrons are not easily available to become protonated.
-
The proton NMR spectrum of 2-pyridone gives the chemical shifts shown. (a) Is 2-pyridone aromatic? (b) Use resonance forms to explain your answer to (a). Also explain why the protons at (7.31and...
-
Explain why each compound is aromatic, antiaromatic, or nonaromatic. (a) (b) (c) (d) (e) (f) (g) (h) N: isoxazole 1.3-thiazole pyran pyrylium ion y-pyrone 1.2-dihydropyridine NH2 N:
-
The following account balances are taken from the ledger of Maxwell Limited on 31 December 2018, the end of its fiscal year: Maxwell Limited Trial Balance As on 31 December 2018 S'm S'm Investment...
-
A researcher wishes to assess whether vitamin C is effective in the treatment of colds. To evaluate this hypothesis, the researcher decides to conduct a 2-year experimental study. The researcher...
-
Cullumber Co. acquired 25% of the 520,000 shares of outstanding common stock of Oriole Inc. on December 31, 2025. The purchase price was $3,913,000. Oriole declared and paid $1.20 per share cash...

Study smarter with the SolutionInn App


