2-Brorno-6, 6-dimethylcyclohexanone gives 2, 2-dirnethylcyclopentane- carboxylic acid on treatment with aqueous NaOH followed by acidification, a process
Question:
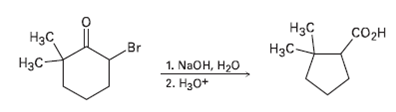
2-Brorno-6, 6-dimethylcyclohexanone gives 2, 2-dirnethylcyclopentane- carboxylic acid on treatment with aqueous NaOH followed by acidification, a process called the Favorskii reaction. Propose a mechanism
Transcribed Image Text:
Нзс Нас Соон Br 1. NaOH, H20 2. На0+ Нас H3C-
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (16 reviews)
H3C H3C 0 OH ...View the full answer

Answered By

Chiranjib Thakur
I have no tutoring experience yet, but I can share my skills and knowledge gained from my education and work experiences. I have been a CPA since 2012 with 6 years of work experience in internal auditing and 4 years of work experience in accounting at the supervisory level.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Propose a mechanism for the reaction of benzoic acid with oxalyl chloride. This mechanism begins like the thionyl chloride reaction, to give a reactive mixed anhydride. Nucleophilic acyl substitution...
-
A compound reacts with methylmagnesium bromide followed by acidification to form the product with the following 1H NMR spectrum. Identify the compound. 6 (ppm) 2 frequency
-
A carboxylic acid has two oxygen atoms, each with two nonbonding pairs of electrons. (a) Draw the resonance forms of a carboxylic acid that is protonated on the hydroxyl oxygen atom. (b) Compare the...
-
Write the Comparison for Legal Factors of China and South Africa
-
Assume that a manager becomes the leader of a division in which the vast majority of the workers are under 25, such as a restaurant chain. Would you recommend that the leader get some body piercing...
-
Lillian purchased a guitar from Smash Music Stores. It regularly sold for $670, but was on sale at 10% off. She paid 8% tax. She bought it on the installment plan and paid 15% of the total cost with...
-
What graphical tool enables us to delineate what is within the scope of a system, product, or services boundaries?
-
For the year ended December 31, 2017 Savory Inc. (Savory) reported net income of $750,000. On December 31, 2016, Savory had the following capital stock outstanding: Preferred shares, no par, $5...
-
The audited accounts of Wilo Ltd . for the year - end 3 1 December 2 0 2 2 , show a loss of $ 6 , 8 0 0 , 0 0 0 after charging the following: $Depreciation 1 , 7 5 0 , 0 0 0 Legal fees 1 , 6 0 0 , 0...
-
What is the major product formed in the following reaction? HBr A) B) Br Br D) C) Br Br
-
Identify the missing reagents af in the following scheme: Br "o2 "
-
In plants, terpenes are biosynthesized by a pathway that involves loss of CO2 from 3-phosphornevalonate 5-diphosphate to yield isopentenyl diphosphate. Use curved arrows to show the mechanism of this...
-
Each month, the owner of Caf Gardens restaurant records y = monthly total sales receipts and x = amount spent that month on advertising, both in thousands of dollars. For the first four months of...
-
What are the interesting objects you have learned about analyzing financial data? Which ratio would you relyon as an investor? as a business manager?
-
Why are financial markets essential for a healthy economy and economic growth? What are financial institutions? Describe each and give an example of who may use them: Investment banks Commercial...
-
Andy purchased a 6% coupon rate commonwealth Government Securities (CGS), which mature in 2022 June. Assume the par value is 100, and YTM is 5% P.A. Calculate the CGS price when Andy purchased this...
-
On January 1, 2009, Bruno owed $8,384 to his friend Alexandra, who was kind enough not to charge Bruno any interest. Each month during 2009, Bruno paid Alexandra some of the money he owed. If Bruno...
-
What role do institutions and governance systems play in fostering an environment conducive to empowerment, and how can these structures be reimagined to better serve marginalized populations ?
-
Well-rounded sediment particles indicate (a) a quiet, low-energy environment. (b) mechanical weathering. (c) glacial deposits. (d) long transport distances.
-
Design and describe an application-level protocol to be used between an automatic teller machine and a bank's centralized computer. Your protocol should allow a user 's card and password to be...
-
Palladium crystallizes with a face-centered cubic structure. It has a density of 12.0 g/cm 3 , a radius of 138 pm, and a molar mass of 106.42 g/mol. Use these data to calculate Avogadros number.
-
Draw a lewis structure for each of the following species. Show all unshared pairs and the formal charges, if any. Assume that bonding follows the octet rule in all cases. (a) NH3 ammonia (b) [NH4]+...
-
Predict the approximate bond angles in each of the following molecules. (a) BeH2 (b) (c) :Cl Si HCC CH (Give H C C and C) _C-C angles. allene
-
The percent s character describes the hybridization of an orbital. For example, an ,sp3 orbital has 25%s character. Given the bond angles in each case, calculate the percent s character of (1) the...
-
What is the balance in Work in Process Inventory at the end of each month? Work in Process Inventory April 30 $ May 31 A 10,140 15,950 June 30 $ 10,450
-
Sako Company s Audio Division produces a speaker used by manufacturers of various audio products. Sales and cost data on the speaker follow: Selling price per unit on the intermediate market $ 6 0...
-
Vulcan Company's contribution format income statement for June is as follows: Vulcan Company Income Statement For the Month Ended June 30 Sales Variable expenses Contribution margin Fixed expenses...

Study smarter with the SolutionInn App


