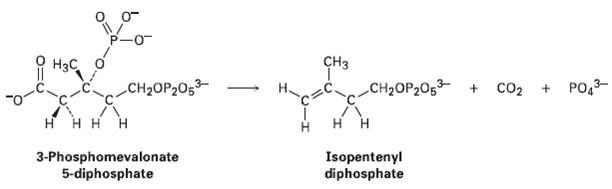
In plants, terpenes are biosynthesized by a pathway that involves loss of CO2 from 3-phosphornevalonate 5-diphosphate to
Question:
In plants, terpenes are biosynthesized by a pathway that involves loss of CO2 from 3-phosphornevalonate 5-diphosphate to yield isopentenyl diphosphate. Use curved arrows to show the mechanism of this reaction.
Transcribed Image Text:
CHз Нас, PO,3- CH2OP205 + co2 + CH2OP2O5³- н нн н ннн 3-Phosphomevalonate 5-diphosphate Isopentenyl diphosphate
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 81% (16 reviews)
OO HH CHOP 05 HH 3Ph...View the full answer

Answered By

Allan Simiyu
I am an adroit Writer. I am a dedicated writer having worked as a writer for 3 years now. With this, I am sure to ace in the field by helping students break down abstract concepts into simpler ideas.
5.00+
8+ Reviews
54+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Use curved arrows to show how calcium carbide reacts with water to give acetylene.
-
Use curved arrows to show how calcium carbide reacts with water to give acetylene.
-
Use curved arrows to show the movement of electrons in each of the following reaction steps: a. b. c. d. Br O: " CH,COH + H-O-H CH,COH + H2O - CH3 CH3 CH CH3 CH
-
Write a report on Home and Automobile: These are two of the most important financial purchases we will make. These decisions, especially housing, will affect much of your ability to meet your...
-
What actions might a leader take to demonstrate that his or her interest in diversity goes beyond rhetoric?
-
Reggie needs a quick x-dollar loan, just until his next payday in two weeks to take advantage of a sale on ski equipment. The bank would take too long in paperwork, so he goes to a pawnshop. The...
-
Why do Modes and States have a critical role in SE problem-solvingsolution development?
-
Winans Company uses the lower-of-cost-or-market method, on an individual-item basis, in pricing its inventory items. The inventory at December 31, 2013, included product X. Relevant per-unit data for...
-
The Food Safety Modernization Act was passed into law in January, 2011. It was a huge reform of US food safety laws, aimed at improving the safety of our national food supply, and taking a proactive...
-
Read the Rohm and Haas case case and then answer the following questions: 1. What should Joan Macey do? a. Specifically with respect to pricing? b. Specifically with respect to distribution? 2. What...
-
2-Brorno-6, 6-dimethylcyclohexanone gives 2, 2-dirnethylcyclopentane- carboxylic acid on treatment with aqueous NaOH followed by acidification, a process called the Favorskii reaction. Propose a...
-
Propose a structure for a compound C 6 H 12 O 2 that dissolves in dilute NaOH and shows the following 1 H NMR spectrum: 1.08 (9 H, singlet), 2.2 (2 H, singlet), and 11.2 (1 H, singlet).
-
Selected transactions for Betz Company during July of the current year are listed in Problem 5-1A. Instructions Journalize the entries to record the transactions of Betz Company for July using the...
-
1.Assume that the equilibrium wage rate is 50AED in the labour market. 2.Discuss what happens if the government introduces a minimum wage of 30AED. 3.Discuss what happens if the government introduces...
-
h. A U.S. Treasury note with a face value of $1000 pays interest semiannually at matures in three years. If the coupon rate is 4% and the bond equivalent yield is 5%, find the price of the note.
-
Consider three incomes: $35 000, $65 000, and $130 000. Determine the maximum RRSP contribution and the tax benefit at each income level. What would be an equivalent TFSA contribution? Next project...
-
DJT, inc. is considering a new investment opportunity that will require an initial investment of $5,951,000.00. The estimated cash flow for year 1 is $1,000,000, for year 2 is $1,500,000, and...
-
What did they do to accelerate analysis and results in general electric case?
-
What is the name of the geologic process that involves dumping sediment at a new location?
-
Use nodal analysis to determine voltages v1, v2, and v3 in the circuit Fig. 3.76. Figure 3.76 4 S 3i, 2 A 4A
-
Polonium crystallizes with a simple cubic structure. It has a density of 9.3 g/cm 3 , a radius of 167 pm, and a molar mass of 209 g/mol. Use these data to calculate Avogadros number (the number of...
-
Consider the resonance structures for the carbonate ion. (a) How much negative charge is on each oxygen of the carbonate ion? (b) What is the bond order of each carbon-oxygen bond in the carbonate...
-
Orbitals with l : 3 are called"f orbitals. (a) How many energetically equivalent f orbitals are there? (b) In [vhat principal quantum level do f orbitals flrst appear? (c) How many nodes does a 5f...
-
The principles for predicting bond angles do not permit a distinction between the following two conceivable forms of ethvlene. The dipole moment of ethylene is zero. Does this experimental fact...
-
How do advanced deadlock detection algorithms operate within large-scale systems, and what are the trade-offs between computational complexity and the speed of deadlock resolution ?
-
Provide an example of typical consumers of energy in real life. Discuss their motives for energy consumption based on economic behavioral models.
-
How does the concept of a "deadlock-free" system align with the practical realities of resource management in operating systems, and what are the potential drawbacks of striving for complete deadlock...

Study smarter with the SolutionInn App


