2-Methylpentanc (C 6 H 14 ) has the mass spectrum shown. Which peak represents M + ?...
Question:
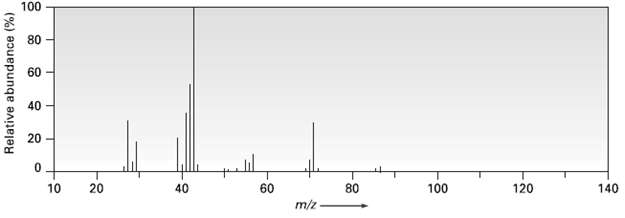
2-Methylpentanc (C6H14) has the mass spectrum shown. Which peak represents M+? Which is the base peak? Propose structures for fragment ions of m/z = 71, 57, 43, and 29. Why does the base peak have the mass it does?
Transcribed Image Text:
100 80 20 120 40 10 140 20 60 80 100 m/z Relative abundance (%) 20 8 유
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (10 reviews)
CHCHCHCHCH mz 71 CH3 CH3CHCHCHCH3 2Methylpentane k ...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The following mass spectrum is for octane. a) Which peak represents the molecular ion? b) Which peak is the base peak? c) Draw the structure of the fragment that produces the base peak. 100 80 60 40...
-
Propose structures and fragmentation mechanisms corresponding to ions with m/z 57 and 41 in the mass spectrum of 4-methyl-1-hexene. onization (loss of a electron) mlz 57 m/z 41
-
Propose reasonable fragmentation mechanisms that explain why The EI mass spectrum of benzoic acid shows major peaks at m/z = 105 and m/z = 77.
-
Solar Energy Corp. has $4 million in earnings with four million shares outstanding. Investment bankers think the stock can justify a P/E ratio of 21. Assume the underwriting spread is 5 percent. What...
-
In the United States, the incomes of specialists such as heart surgeons can easily triple the incomes of primary care practitioners. Use the Five Forces to offer explanation for this disparity. Can...
-
(a) The PH3 molecule is polar. Does this offer experimental proof that the molecule cannot be planar? Explain. (b) It turns out that ozone, O3, has a small dipole moment. How is this possible, given...
-
What is the difference between social media discrimination and negligent hiring?
-
By ordering unnecessary products at inflated prices, the purchasing manager of XYZ Company defrauded his employer of over $40,000 over a two-year period. How could you have detected this fraud?
-
2) If you strike a pool ball with just the right amount of back spin, it will come to a perfect stop. (a) What is the appropriate relationship between the initial velocity, vi, and the initial...
-
Economy A and Economy B are similar in every way except that in Economy A, 70percent of aggregate expenditure is sensitive to changes in the real interest rate and in economy B, only 50 percent of...
-
Propose structures for compounds that fit the following data: (a) A ketone with M = 86 and fragments at m/z = 71 and m/z = 43 (b) An alcohol with M + = 88 and fragments at m/z = 73, m/z = 70, and m/z...
-
Assume that you are in a laboratory carrying out the catalytic hydrogenation of cyclohexane to cyclohexane. How could you use a mass spectrometer to determine when the reaction is finished?
-
Define the following terms: magnetic induction, magnetic field, magnetic susceptibility, and magnetic permeability.
-
If Gaga's sales are $19391, variable expenses are $2231, and the fixed expenses total $2806 per period, by what dollar amount will net operating income change if sales are expected to increase by...
-
A copper kettle weighs approximately 0.98 kg. Suppose this kettle contains 800 mL of water at a temperature of 21.5C. How much heat (in kJ) would be required to raise the temperature of the water to...
-
A corporate bond with a $1,000 face value pays a $50 coupon semi-annually. The bond will mature in ten years, and has a nominal yield to maturity of 9 percent. What is the price of the bond?
-
The following condensed balance sheet is for the partnership of Ludolf, Sambal, and Urad, who share profits and losses in the ratio of 6 : 2 : 2 , respectively: Cash $ 4 1 , 0 0 0 Liabilities $ 2 9 ,...
-
Two charged spheres are held 1.0 m apart. If the left sphere has charge 4x10 -6 C and the right sphere has charge 7x10 -6 C, how much force do the two spheres push on each other? Your answer should...
-
A block initially at rest is released on an inclined surface. The block slides down, compressing a spring at the bottom of the incline; there is friction between the surface and the block. Consider...
-
CdF2 (s) Cd+ (aq) + 2 F- (aq) 1. A saturated solution of CdF2 is prepared. The equilibrium in the solution is represented above. In the solution [Cd+] eq = 0.0585 M and [F-] eq = 0.117 M. a....
-
The osmotic pressure of a solution containing 2.10 g of an unknown compound dissolved in 175.0 mL of solution at 25 C is 1.93 atm. The combustion of 24.02 g of the unknown compound produced 28.16 g...
-
Show how you might synthesize the following compounds, using acetylene and any suitable alkyl halides as your starting materials. If the compound given cannot be synthesized by this method, explain...
-
Show how you would synthesize each compound, beginning with acetylene and any necessary additional reagents. (a) prop-2-yn-1-ol (propargyl alcohol) H----C === C--- CH2OH (b) (c) (d) hept-2-yn-4-ol OH...
-
Show how you would synthesize 2-phenylhex-3-yn-2-ol, starting with acetophenone (PhCOCH3) and any other reagents you need. ("2-ol" means there is an OH group on C2.)
-
Proposals to increase inheritance taxes are also under discussion. These are taxes on the amount of money that a person (an heir) inherits. What would the likely effect on the labor supply of heirs...
-
In 2011, a new program was put in place that allowed Christmas trees to be taxed (initially $1 per tree), with funds going to the Christmas Tree Promotion Board. The CTPB would use the funds to...
-
W W2 W3 W4 f f f f Consider the following two-sided matching problem. ff2 f3 f Wi W3 WA f2 fa fa fa fa fa f2 fi fa f2 fi f2 102 W3 WA W2 W3 W2 W2 Wi Wa Wa Wi W3 Compute the W-DA outcome. Compute the...

Study smarter with the SolutionInn App


