The following mass spectrum is for octane. a) Which peak represents the molecular ion? b) Which peak
Question:
a) Which peak represents the molecular ion?
b) Which peak is the base peak?
c) Draw the structure of the fragment that produces the base peak.
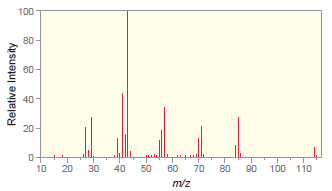
Transcribed Image Text:
100 80 60 40 20 110 10 20 30 40 50 60 70 80 90 100 m/z Relative Intensity
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 56% (16 reviews)
a The molecular io...View the full answer

Answered By

Nandana Wijayarathna
I am a highly experienced writer in several areas,
Business management
Information technology
Business administration
Literature
Biology
Environmental science
History
4.50+
161+ Reviews
399+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
2-Methylpentanc (C 6 H 14 ) has the mass spectrum shown. Which peak represents M + ? Which is the base peak? Propose structures for fragment ions of m/z = 71, 57, 43, and 29. Why does the base peak...
-
Calculate the mass of octane, C8H18(l), that must be burned in air to evolve the same quantity of energy as produced by the fusion of 1.0 g of hydrogen in the following fusion reaction: Assume that...
-
Use Figure 24-15 to suggest which type of liquid chromatography you could use to separate compounds in each of the following categories: (a) Molecular mass < 2 000, soluble in octane (b) Molecular...
-
In Exercises 7980, find the value of y if the line through the two given points is to have the indicated slope. (3, y) and (1, 4), m = -3
-
Write an essay about a "Magazine" focusing on the demographics, describing the niches the magazine is trying to reach. For this the chosen magazine is: Elle magazine. Some demographics that should be...
-
Honda Motor Company is considering offering a $2500 rebate on its minivan, lowering the vehicles price from $21,000 to $18,500. The marketing group estimates that this rebate will increase sales over...
-
Smartpages.com issued stock during 2008 and reported the following on its balance sheet at December 31, 2008: Journalize the company's issuance of the stock for cash. Common stock, $0.25 par value...
-
Determine the force in members BC, HC, and HG. After the truss is sectioned use a single equation of equilibrium for the calculation of each force. State if these members are in tension or...
-
MD Tech is currently not paying any dividends. Analysts expect that MD Tech will pay dividends for the first time in five years from today. They believe that the first dividend (D5) will be $2.0 and...
-
An aircraft engine is fitted with a single-sided centrifugal compressor. The aircraft flies with a speed of 230 m/s at an altitude where the pressure is 0.23 bar and temperature 217 K. The intake...
-
Propose a molecular formula that fits the following data. a) A hydrocarbon (C x H y ) with a molecular ion peak at m/z = 66 b) A compound that absorbs IR radiation at 1720 cm -1 and exhibits a...
-
Calculate the HDI for each molecular formula. a) C 4 H 6 b) C 5 H 8 c) C 40 H 78 d) C 72 H 74 e) C 6 H 6 O 2 f) C 7 H 9 NO 2 g) C 8 H 10 N 2 O h) C 5 H 7 Cl 3 i) C 6 H 5 Br j) C 6 H 12 O 6
-
How do mining experts differentiate between mineral reserves and mineral resources?
-
What is the difference between bivariate regression and multiple regression?
-
Suppose that the price of an asset follows a Brownian motion : \[d S=\mu S d t+\sigma S d z\] (a) What is the stochastic process for \(S^{n}\) ? (b) What is the expected value for \(S^{n}\) ?
-
What is dividend?
-
Do you think it is possible to have win-win negotiations? Why or why not?
-
Differentiate between Type I and Type II errors.
-
Identify two points in the audit process when analytical procedures are required by GAAS, and explain why they are considered necessary.
-
Planning: Creating an Audience Profile; Collaboration: Team Projects. Compare the Facebook pages of three companies in the same industry. Analyze the content on all available tabs. What can you...
-
Is there an unbranched alkane containing 23 hydrogen atoms? If so, give its structural formula; if not, explain why not.
-
In the structure of 4- isopropy 1-2,4,5-trimethylheptane (Problem 2.9) (a) Identify the primary, secondary, tertiary, and quaternary carbons. (b) Identify the primary, secondary, and tertiary...
-
In the structure of 4- isopropy 1-2,4,5-trimethylheptane (Problem 2.9) (a) Identify the primary, secondary, tertiary, and quaternary carbons. (b) Identify the primary, secondary, and tertiary...
-
Imagine you are the assistant manager of the place in which you currently work. You have been instructed by the manager to organize and facilitate a company team building activity to promote...
-
Analyze key change management principles. The organization is having difficulties and frustration regarding outdated technology. Explain the purpose and discipline of change management. Outline a...
-
Organizational chart (project team) for renovating Santa Rita group home , Responsibility matrix Work Breakdown Structure, Communications plan, Project charter and Detailed explanations explaining...

Study smarter with the SolutionInn App


