A cumulene is a compound with three adjacent double bonds. Draw an orbital picture of a cumulene.
Question:
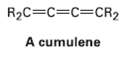
A cumulene is a compound with three adjacent double bonds. Draw an orbital picture of a cumulene. What kind of hybridization do the two central carbon atoms have? What is the geometric relationship of the sub-stituents on one end to the sub-stituents on the other end? What kind of isomerism is possible? Make a model to help see theanswer.
Transcribed Image Text:
R2C=C=C=CR2 A cumulene
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (8 reviews)
sp2 R R spsp o bond a bonds sp sp o bond bonds sp spsp o bond sp R3 R4 This simple...View the full answer

Answered By

ANDREW KIPRUTO
Academic Writing Expert
I have over 7 years of research and application experience. I am trained and licensed to provide expertise in IT information, computer sciences related topics and other units like chemistry, Business, law, biology, biochemistry, and genetics. I'm a network and IT admin with +8 years of experience in all kind of environments.
I can help you in the following areas:
Networking
- Ethernet, Wireless Airmax and 802.11, fiber networks on GPON/GEPON and WDM
- Protocols and IP Services: VLANs, LACP, ACLs, VPNs, OSPF, BGP, RADIUS, PPPoE, DNS, Proxies, SNMP
- Vendors: MikroTik, Ubiquiti, Cisco, Juniper, HP, Dell, DrayTek, SMC, Zyxel, Furukawa Electric, and many more
- Monitoring Systems: PRTG, Zabbix, Whatsup Gold, TheDude, RRDtoo
Always available for new projects! Contact me for any inquiries
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Draw an orbital picture of furan to show how the molecule isaromatic. Furan :O:
-
Draw an orbital picture of thiazole. Assume that both the nitrogen and sulfur atoms are sp2-hyhridized, and show the orbitals that the lone pairs occupy.
-
What kind of hybridization do you expect for each carbon atom in the followingmolecules? (a) Propane, CH3CH2CH3 (b) 2-Methylpropene, CH CH3C=CH2 (c) 1-Butene 3 yne, H2C=CH-C=CH (d) Acetic acid, CHC
-
The balance sheet for the Heir Jordan Corporation follows. Based on this information and the income statement in the previous problem, supply the missing information using the percentage of sales...
-
You are the Director of the Computer Center for Gaillard College and responsible for scheduling the staffing of the center, which is open from 8 a.m. until midnight. You have monitored the usage of...
-
Determine v1 and v2 in the circuit in Fig. 3.71. Figure 3.71 8 3 A 12 +lo 1(2 12 V 4 5
-
When preparing departmental contribution statements of financial performance, all direct expenses are controllable by department managers. Discuss, giving examples.
-
Update the trial balance for Kylers Moving Co. (Figure) for January 31, 201X. Adjustment Data to Update Trial Balance a. Insurance expired, $550. b. Moving supplies on hand, $700. c. Depreciation on...
-
Liquidity Ratios Debt Management Ratios Profitability Ratios Market Value Ratio Current Ratio Days Sales in Receivables Debt/Equity Long Term Debt/Capital Return on Assets Return on Equity Net Profit...
-
Your parents are considering investing in Apple Inc. common stock. They ask you, as an accounting expert, to make an analysis of the company for them. Financial statements of Apple are presented in...
-
Terminal alkynes react with Br2 and water to yield bromo ketones. For example: Propose a mechanism for the reaction. To what reaction of alkenes is the processanalogous? -CECH Br2, H20 CH2Br
-
Reaction of acetone with D 3 O + yields hexadeuterioacetone. That is, all the hydrogen?s in acetone are exchanged for deuterium. Review the mechanism of mercuric ion?catalyzed alkyne hydration, arid...
-
A steam power plant has an instantaneous net power delivery of 462 MW with a heat rate of 11,827 Btu/kWh. For this operating condition determine, a. The boiler heat transfer rate (MBtu/hr) b. The...
-
1.How does the positivist school differ from the classical school of criminology (e.g., think about the concept of predestination)? Describe the tenets of each and how they differ. Which of these...
-
1. The yield point data necessary to experimentally construct a yield surface for the particle reinforced aluminum 6092/17.5p-W is provided below. These data were acquired using axial- torsional...
-
The Objective is to develop a strategic leadership development plan. Throughout this course, we have examined in depth the process of strategic leadership and the various elements that make...
-
Examine Table 2.1. Describe the biological reason row 1 and row 6 of the data in th table differ from each other. Limit response to 40 words maximum. genotype PO B P 10 B P " O* _Z+ _P* / * _O+ _g*...
-
The energy E of the electron in a hydrogen atom can be calculated from the Bohr formula: = E Ryn 2 In this equation Ry stands for the Rydberg energy, and n stands for the principal quantum number of...
-
If you know the names of the starting batters for a baseball team, what is the probability of randomly selecting a batting order and getting the order that is used in the beginning of the game?...
-
For each of the following transactions, indicate whether it increases, decreases, or has no effect on the following financial ratios: current ratio, debt-to-equity ratio, profit margin ratio, and...
-
Determine whether each redox reaction is spontaneous. (a) Fe(s) + Mg2+ (aq) (b) Fe(s) + Pb+ (aq) Fe2+ (aq) + Mg(s) 2+ Fe+ (aq) + Pb(s)
-
(a) Draw the structure of cis-CH3-CH=CH-CH2CH3 showing the pi bond with its proper geometry. (b) Circle the six coplanar atoms in this compound. (c) Draw the trans isomer, and circle the coplanar...
-
In pent-2-yne (CH3CCCH2CH3) there are four atoms in a straight line. Use dashed lines and wedges to draw a threedimensional representation of this molecule, and circle the four atoms that are in a...
-
Which of the following compounds show cis-trans isomerism? Draw the cis and trans isomers of the ones that do. (a) CH3CH=CHCH3 (b) CH3-C¡C-CH3 (c) CH2=C(CH3)2 (d) (e) (f) CH3-CH=N-CH3...
-
The cash account for the Justice Company at June 30, 20X8 indicated a balance of $5900. The bank statement indicated a balance of $5800 on June 30, 20X8. Comparing the bank statement and the...
-
The cash account for the Justice Company at June 30, 20X8 indicated a balance of $5900. The bank statement indicated a balance of $5800 on June 30, 20X8. Comparing the bank statement and the...
-
The following information is provided for Roberts Company as of June 30, 20X8. 1. Bank Balance: $8638 2. Deposits in transit: $2200 3. Service charges: $40 4. Book Balance: $9700 5. First Bank...

Study smarter with the SolutionInn App


