What kind of hybridization do you expect for each carbon atom in the followingmolecules? (a) Propane, CH3CH2CH3
Question:
What kind of hybridization do you expect for each carbon atom in the followingmolecules?
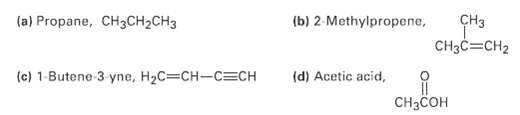
Transcribed Image Text:
(a) Propane, CH3CH2CH3 (b) 2-Methylpropene, CHз CH3C=CH2 (c) 1-Butene 3 yne, H2C=CH-C=CH (d) Acetic acid, CHзCсон
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 52% (17 reviews)
a sp3 sp3 sp3 CH3C...View the full answer

Answered By

Muhammad Haroon
More than 3 years experience in teaching undergraduate and graduate level courses which includes Object Oriented Programming, Data Structures, Algorithms, Database Systems, Theory of Automata, Theory of Computation, Database Administration, Web Technologies etc.
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
What kind of hydribization do you expect for each carbon atom in the followingmolecules? CH- (b) H. C-H (a) 1 "CH-CH CH2 C=C H2N H. Procaine Vitamin C (ascorbic acid)
-
What hybridization do you expect for the atom indicated in red in each of the following species? (a) CH3CO2- (b) PH4+ (c) AlF3 (d) H2C==CH-CH2+
-
What dividends do you expect for Goodman Industries stock over the next 3 years if you expect the dividend to grow at the rate of 5% per year for the next 3 years? In other words, calculate D1, D2,...
-
Would individual mandates for health insurance be more or less burdensome to the poor than employer mandates? Would lower-income groups be wise to favor one plan over the other?
-
Art Funkel started his incorporated medical practice on June 1, 2016. He immediately made an Selection for the corporation. Art would like the corporation to adopt a tax year ending May 31 so that a...
-
One disadvantage of GIF is that the image must include the color palette, which increases the file size. What is the minimum image size for which an 8-bit-wide color palette breaks even? Now repeat...
-
Calculate the efficiency of the collector described in Problem 7.1 on March 1 at a latitude of \(40^{\circ} \mathrm{N}\) between 11 and 12 a.m. Assume that the total horizontal insolation is \(450...
-
The manager of the aerospace division of General Aeronautics has estimated the price it can charge for providing satellite launch services to commercial firms. Her most optimistic estimate (a price...
-
Good Dog Enterprises sells goods with a warranty. At December 31, 2021 Good Dog has a warranty liability of $2 million and taxable income of $75 million. At December 31, 2020 Good Dog reported a...
-
Suppose this morning SPB Ltd., a solar panel and battery manufacturing company, paid a dividend of $5 per share. At 10 a.m. SPB announced its latest earnings results (which were unexpectedly good)...
-
Convert the following molecular formulas into line-bond structures that are sonsistent with valence rules: (a) C3H8 (b) CH5N (c) C2H6O (2 possibilities) (d) C3H7Br (2 possibilities) (e) C2H4O (3...
-
What is the shape of benzene, and what hydribization do you expect for eachcarbon? C=c H-C - Benzene C-C I.
-
True or False: In a large corporation it is usually better for the central management to make decisions rather than divisional managers, because the central management has access to a wider range of...
-
How would increasing or decreasing a company's cash flows affect their NPV?
-
Often the normative objective function of the financial manager and social responsibility are incompatible. Social responsibility means giving back to the community and the normative object function...
-
search about Toronto stock exchange what is it? what are the functions? how shares are listed etc. Then compare it with New York Stock Exchange. What is the difference and what are the similarities?
-
With a lower revenue growth target, would HH be able to avoid a cash deficiency? Discuss what other measures the Browns might take in order to approach the cash level that Maggie considers ideal,...
-
If the stock price of Coca Cola (KO) on the Yahoo.com website reads like this as of 4/12/2013: A) What was the closing price of the stock for the previous day? B) What is the current price of the...
-
What is an important advantage of a Pareto chart over a pie chart?
-
Presented below are income statements prepared on a LIFO and FIFO basis for Kenseth Company, which started operations on January 1, 2024. The company presently uses the LIFO method of pricing its...
-
Calculate the vapor pressure of n-butane as a function of temperature using the Peng-Robinson equation of state. Compare your results with (a) Literature values (b) Predictions using the...
-
Explain whether each pair of models represents isomers or the same compound. (All represent compounds with the formula C7H16.) Draw structures for each compound represented by the models.
-
Explain whether each pair of models represent isomers or the same compound. Draw structures for each compound represented by the models.
-
The following models represent three isomers of C6H4Cl2. Explain which of these compounds does not have a dipole moment.
-
Solve the initial value problem. dy =3e3t sin (e3t-27), y(In 3)=0 dt The solution is y =
-
Solve by factoring. 5x2-16x+12=0
-
Find f-1 (x) for f (x) = 15 + 12x.

Study smarter with the SolutionInn App


