(a) Derive the equation relating the molality (m) of a solution to its molarity (M) Where d...
Question:
(a) Derive the equation relating the molality (m) of a solution to its molarity (M)
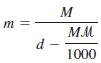
Where d is the density of the solution (g/mL) and m is the molar mass of the solute (g/mol).
(b) Show that, for dilute aqueous solutions, m is approximately equal to M.
Transcribed Image Text:
M m MM d 1000
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (14 reviews)
a If we assume 1 L of solution then we can calculate the mass of solution from its density ...View the full answer

Answered By

Mercy Kangai
I provide creative and detailed administrative, web search, academic writing, data entry, Personal assistant, Content writing, Translation, Academic writing, editing and proofreading services. I excel at working under tight deadlines with strict expectations. I possess the self-discipline and time management skills necessary to have served as an academic writer for the past seven years. I can bring value to your business and help solve your administrative assistant issues. I have extensive experience in marketing and small business management.
4.80+
27+ Reviews
86+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
One practical way to measure magnetic field strength uses a small, closely wound coil called a search coil. The coil is initially held with its plane perpendicular to a magnetic field. The coil is...
-
Derive an equation similar to the Henderson-Hasselbalch equation relating the pOH of a buffer to the of its base component.
-
A conveyor drive system to produce translation of the load is shown in Figure. The reducer is a gear pair that reduces the motor speed by a factor of 10:1. The motor inertia is I = 0.002 kg m2. The...
-
List four components and four guidelines that the J. Crew mission statement fails to exhibit. Write a new and improved mission for J. Crew.
-
You purchased your house 5 years ago for $110,000 and based on recent appraisals it can be sold today for $141,000. What effective annual rate of return did you earn?
-
Considering all the above, what screens/factors might you suggest to narrow down the set of most desirable projects? What criteria would you use to evaluate the projects on these various factors? Do...
-
What problems to be aware of when accessing social media sites?
-
Joseph is the named insured under a Homeowners 3 policy (special form) with a liability limit of $100,000 per occurrence and a $1000 limit for medical payments to others. For each of the following...
-
Variances, Entries, and Income Statement A summary of Blake Company's manufacturing variance report for June follows. Total Standard Costs (7,600 units) Actual Costs (7,600 units) Variances Direct...
-
Jillian Limited ( JL) issued a financial instrument with the following terms: A face value of $ 100. Not secured by any assets of the entity (unsecured). Redeemable in cash at the option of the...
-
Using Henry's law and the ideal gas equation to prove the statement that the volume of a gas that dissolves in a given amount of solvent is independent of the pressure of the gas.
-
At 298 K, the osmotic pressure of a glucose solution is 10.50 atm. Calculate the freezing point of the solution. The density of the solution is 1.16 g/mL.
-
Have you ever walked down a street in a tourist area and seen a lot of restaurants with similar menus? Explain why the restaurants might be considered to be operating in monopolistic competition.
-
What steps should an auditor take to minimise the risks of litigation?
-
Most firms do not want other companies to copy their successful business model. However, the shoe retailer TOMS is not your typical retailer. Although many organizations try to incorporate...
-
What are the two primary modes of reporting audit results?
-
How does the internal auditor prepare to write the audit report?
-
What are two common types of flowcharting techniques?
-
1. Discuss how IKEAs approach has helped you to better understand the debate that exists between standardization compared with adaptation for international markets? 2. Compare and contrast two...
-
1. Which of the four major types of information systems do you think is the most valuable to an organization? 2. How do you critically associate the ideas of business agility and business efficiency...
-
Ignoring double-bond stereo chemistry, what products would you expect from elimination reactions of the following alkyl halides? Which will be the major product in eachcase? Br CH CH CI CH (a) (b) Br...
-
What alkyl halides might the following alkenes have been madefrom? (b) C CH (a) (a) CH CHH2H2%3DCH2 "CH
-
What stereo chemistry do you expect for the alkene obtained by E2 elimination of (1R, 2R)-1, 2-dibromo-1, 2-diphenylethane? Draw a Newman projection of the reacting conformation?
-
Sheridan Co. leased machinery from Young, Inc. on January 1, 2025. The lease term was for 8 years, with equal annual rental payments of $5,500 at the beginning of each year. In addition, the lease...
-
Lasky Manufacturing has two divisions: Carolinas and Northeast. Lasky has a cost of capital of 7.5 percent. Selected financial information (in thousands of dollars) for the first year of business...
-
The Game Zone sells computer and other electronic games. The store has budgeted sales for January Year 2 as indicated in the following table. The company expects a 4% increase in sales for the month...

Study smarter with the SolutionInn App


