(a) For 1.0 mol of a monatomic ideal gas taken through the cycle in Figure, where V1...
Question:
(a) For 1.0 mol of a monatomic ideal gas taken through the cycle in Figure, where V1 = 4.00V0, what is W/p0V0 as the gas goes from state a to state c along path abc what is ΔEint/p0V0 in going?
(b) From b to c and
(c) Through one full cycle? What is AS in going?
(d) From b to c and
(e) Through one full cycle?
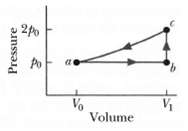
Transcribed Image Text:
Vo Volume Pressure
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
a Work is done only for the ab portion of the process This portion is at constant pressure so the wo...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Fundamentals of Physics
ISBN: 978-0471758013
8th Extended edition
Authors: Jearl Walker, Halliday Resnick
Question Posted:
Students also viewed these Thermodynamics questions
-
For 2.60 moles of a monatomic ideal gas taken through the cycle in Fig 20-24, where V1 = 6.OVO what is W/pOVO as the gas goes from state a to state c along path abc? what is Î Eint / pOVO in...
-
In a heat engine, 3.00 mol of a monatomic ideal gas, initially at 4.00 atm of pressure, undergoes an isothermal expansion, increasing its volume by a factor of 9.50 at a constant temperature of 650.0...
-
As a 1.00-mol sample of a monatomic ideal gas expands adiabatically, the work done on it is - 2 500 J. The initial temperature and pressure of the gas are 500 K and 3.60 atm. Calculate (a) The final...
-
Which of the theoretical approaches are the most useful in explaining your organisations current IHRM policies and practices?
-
For the statement of Problem 1: (a) Determine the dollar value of the consumer surplus before and after the imposition of the tariff. (b) Of the increase in the revenue of producers with the tariff...
-
Map the object-oriented database design for the DreamHome case study produced in Exercise 27.25 to the ODMG ODL.
-
The boundary condition corresponding to the free end of a bar in longitudinal vibration is given by a. \(u(0, t)=0\) b. \(\frac{\partial u}{\partial x}(0, t)=0\) c. \(A E \frac{\partial u}{\partial...
-
Singleton Supplies Corporation (SSC) manufactures medical products for hospitals, clinics, and nursing homes. SSC may introduce a new type of X-ray scanner designed to identify certain types of...
-
Inventory, by Product Category Quantity Per Unit Cost Net Realizable Value Tools: Hammers Saws Screwdrivers Paint products: 1-gallon cans Paint brushes 110 $ 5.40 $5.90 240 10.40 9.40 340 2.40 3.00...
-
Answer all parts of Problem 3.15 for the utility function U(x, y) = x0.4y0.6. The marginal utilities are MUx = 0.4 (y0.6/x0.6) and MUy = 0.6 (x0.4/y0.4). In problem 15 a) Is the assumption that more...
-
A 10 g ice cube at 10oC is place in a lake whose temperature is 15oC. Calculate the change in entropy of the cube lake system as the ice cube comes to thermal equilibrium; with the lake. The...
-
A mixture of 1773 g of water and 227 g of ice is in an initial equilibrium state at 0.000oC. The mixture is then, in a reversible process, brought to a second equilibrium state where the water-ice...
-
Peter has $30,000 to invest in a mutual fund whose annual returns are normally distributed with a mean of 5% and standard deviation of 4.2%. a. Use Analysis ToolPak or R, both with a seed of 1, to...
-
Safety at Petro-Plus Quick Stop Camilla Conrad has been employed for just over four years as a server at the Petro-Plus Quick Stop( PPQS ) restaurant in Kentville Nova Scotia. PPQS gets extremely...
-
Which aspect of environment law makes it uniquely hard to sue for environmental harm? 1. It is often hard to find a plaintiff willing to sue for environmental damage 2. It is uncommon for entities to...
-
On March 31, a company takes a short position in May futures on 100,000 bu of corn at $4.663/bu. The size of one contract is 5,000 bu. The initial margin is $3,894 per contract and the maintenance...
-
Reviewing your team project and policy paper, I would like for you to express and outline. Think about these questions in responding: - What factors influence how need is determined? - How are...
-
1. List the multiple types of sport facilities and describe how sport managers and event planners can produce revenue for each. Consider the versatility of each facility. 2.Does the type of facility...
-
Two charged particles are placed near the origin of an \(x y z\) coordinate system. Particle 1 carries charge \(+q\) and is on the \(x\) axis at \(x=+d\). Particle 2 carries an unknown charge and is...
-
Is it a breach of fiduciary duty for a director of a real estate investment trust (REIT) negotiating a joint venture on behalf of the REIT with another director for the development of a portfolio of...
-
Two piloted satellites approaching one another, at a relative speed of 0.250 m/s, intending to dock. The first has a mass of 4.00 x 10 3 kg, and the second a mass of 7.50 x 10 3 kg. (a) Calculate the...
-
How fast (as a percentage of light speed) would a star have to he moving so that the frequency of the light we receive from it is 10.0% higher than the frequency of the light it is emitting? Would it...
-
The shock-wave cone created by the space shuttle at one instant during its reentry into the atmosphere makes an angle of 58.0 with its direction of motion. The speed of sound at this altitude is 331...
-
A jet plane flies overhead at Mach 1.70 and at a constant altitude of 950 m. (a) What is the angle a of the shock-wave cone? (b) How much time after the plane passes directly overhead do you hear the...
-
The December 31, Year 4, balance sheet for Vernon Corporation is presented here. These are the only accounts on Vernon's balan sheet. Amounts indicated by question marks (?) can be calculated using...
-
Reyes Manufacturing Company uses a job order cost system. At the beginning of January, the company had one job in process (Job 201) and one job completed but not yet sold (Job 200). Job 202 was...
-
n-1 3.) Given the discrete sequence x(n) = (3) u(n - 1) where u is the unit step sequence, do the following: (SHOW ALL WORK) a.) Compute the discrete time Fourier Transform (DTFT) X(w) of x(n). b.)...

Study smarter with the SolutionInn App


