A gas within a closed chamber undergoes the cycle shown in the p-V diagram of Figure. The
Question:
A gas within a closed chamber undergoes the cycle shown in the p-V diagram of Figure. The horizontal scale is set by Vs = 4.0 m3. Calculate the net energy added to the system as heat during one complete cycle.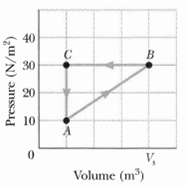
Transcribed Image Text:
40 30 20 10 V, Volume (m) Pressure (N/m)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (10 reviews)
Over a cycle the internal energy is the same at the beginning and end so the heat Q ...View the full answer

Answered By

MICHAEL KICHE
I was employed studypool for the first time in tutoring. I did well since most of my students and clients got the necessary information and knowledge requested for. I always submitted the answers in time and followed the correct formatting in answering eg MLA or APA format,
Again I worked with the writers bay where I did writing and got many clients whom we worked with so closely. They enjoyed every single service I delivered to them. My answers are always correct.
4.70+
13+ Reviews
53+ Question Solved
Related Book For 

Fundamentals of Physics
ISBN: 978-0471758013
8th Extended edition
Authors: Jearl Walker, Halliday Resnick
Question Posted:
Students also viewed these Thermodynamics questions
-
Gas within a chamber passes through the cycle shown in Figure. Determine the energy transferred by the system as heat during process CA it the energy added as heat QAB during process AB is 20.0 J, no...
-
A monatomic ideal gas is taken around the cycle shown in Fig. 20.29 in the direction shown in the figure. The path for process c → a is a straight line in the pV-diagram. (a) Calculate Q, W, and...
-
A heat engine follows the cycle shown in the figure. (a) How much net work is done by the engine in one cycle? (b) What is the heat flow into the engine per cycle? 4.00 atm 1.00 atm 0.200 m 0.800 m3 T
-
Consider the examples of groupthink explained in this chapter and discuss how they can relate to situations you have experienced on your projects.
-
Firm Q operates a cash basis consulting business. In October, Firm Q billed a client for $23,400 of consulting services. In November, the client settled the bill by paying $10,000 cash and...
-
Martinez Ltd. has the following equity accounts at January 1, 2017. Preferred shares outstanding: 2,500 shares ........................ $ 62,500 Common shares outstanding: 4,000 shares...
-
When a suspected drunk driver blows 188 mL of his breath through the fuel-cell breathalyzer described in Section 20.7, the breathalyzer produces an average of 324 mA of current for 10 s. Assuming a...
-
David R. and Ella M. Cole (ages 39 and 38, respectively) are husband and wife who live at 1820 Elk Avenue, Denver, CO 80202. David is a regional sales manager for Wren Industries, a national...
-
Assume the perpetual inventory system is used. Required: Complete the table to determine the cost assigned to ending inventory and cost of goods sold using specific identification. Determine the cost...
-
A storeroom is used to organize items stored in it on N shelves. Shelves are numbered from 0 to N-1. The K-th shelf is dedicated to items of only one type, denoted by a positive integer A[K]....
-
A 20.0 g copper ring at 0.000oC has an inner diameter of D = 2.54000 cm. An aluminum sphere at 100.0oC has a diameter of d = 2.545 08 cm. The sphere is placed on top of the ring (Figure), and the two...
-
Suppose 200 J of work is done on a system and 70.0 cal is extracted from the system as heat. In the sense of the first law of thermodynamics, what are the values (including algebraic signs) of (a) W,...
-
Explain why Hong Kong has experienced faster economic growth than the United States.
-
Why is it important for the project manager to be accurate in the submission of pay applications? What is lost if the amount requested is low? What is the risk if the amount requested is more than...
-
Why is production cost reduction the primary means to deal with cost overruns?
-
Why does the placement of blame have no place in the postproject evaluation process?
-
What advantage does an Earned Value Management System have over more traditional cost-control and schedule-control systems?
-
Current technology gives project managers instant access to cost accounting systems virtually anytime. Why is weekly reporting still the most common cycle for construction cost accounting reports?
-
Stock prices are a central element in a market economy, because they ensure that investment resources flow to their most profitable uses. When occasional bubbles and crashes distort stock prices,...
-
How many years will it take a $700 balance to grow into $900 in an account earning 5%?
-
Diagram a negative reinforcement contingency in the Skinner box.**
-
Make an order-of-magnitude estimate of the electric current that the electric company delivers to a town (Figure P33.64) from a remote generating station. State the data you measure or estimate. If...
-
Consider a series RLC circuit having the following circuit parameters: R = 200 , L = 663 mH, and C = 26.5 F. The applied voltage has an amplitude of 50.0 V and a frequency of 60.0 Hz. Find the...
-
A voltage v = (100 V) sin wt (in SI units) is applied across a series combination of a 2.00-H inductor, a 10.0-F capacitor, and a 10.0- resistor. (a) Determine the angular frequency w0 at which the...
-
Select three services: one high in search attributes, one high in experience attributes, and one high in credence attributes. Specify what product characteristics make them easy or difficult for...
-
The New York City Council is ready to vote on two bills that authorize the construction of new roads in Manhattan and Brooklyn. If the two boroughs join forces, they can pass both bills, but neither...
-
Miss Israel has a chest tube in place for a spontaneous pneumothorax. You note that the water seal chamber is bubbling vigorously. What could cause this? What should you do? You are totaling intake...

Study smarter with the SolutionInn App


