A solution contains two volatile liquids A and B. Complete the following table, in which the symbol
Question:
A solution contains two volatile liquids A and B. Complete the following table, in which the symbol → indicates attractive intermolecular forces.
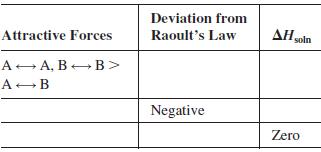
Transcribed Image Text:
Deviation from Attractive Forces Raoult's Law AH soln A A, B B> A-B Negative Zero
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (15 reviews)
The completed table is shown below The first row represents a Case 1 situat...View the full answer

Answered By

Carly Cimino
As a tutor, my focus is to help communicate and break down difficult concepts in a way that allows students greater accessibility and comprehension to their course material. I love helping others develop a sense of personal confidence and curiosity, and I'm looking forward to the chance to interact and work with you professionally and better your academic grades.
4.30+
12+ Reviews
21+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The volatile liquids A and B, for which P * A = 165 Torr and P * B = 85.1. Torr are confined to a piston and cylinder assembly. Initially, only the liquid phase is present. As the pressure is...
-
For a pure substance, the liquid and gaseous phases can only coexist for a single value of the pressure at a given temperature. Is this also the case for an ideal solution of two volatile liquids?
-
A mixture of liquids A and B exhibits ideal behavior. At 84C, the total vapor pressure of a solution containing 1.2 moles of A and 2.3 moles of B is 331 mmHg. Upon the addition of another mole of B...
-
Four of Wands, LLC generated $255,000 in sales during January 2022. Of this amount, 25% was for cash. The remaining 75% of sales were made on account. The February 2022 sales on account were...
-
Clark Paints: The production department has been investigating possible ways to trim total production costs. One possibility currently being examined is to make the paint cans instead of purchasing...
-
Leicht Transfer & Storage provides warehousing services and often purchases pallets from Pallet Central. The companies followed a standard practice for documenting these transactions in which Pallet...
-
As auditor for the Court Company, you decide to use variables sampling to estimate the total cost of an inventory of 1,150 items which has a net book value of \(\$ 260,400\). The auditor decides to...
-
The Rushing Construction Company obtained a construction contract to build a highway and bridge over the Snake River. It was estimated at the beginning of the contract that it would take three years...
-
Required: Deer Lake Incorporated uses a job order cost system with manufacturing overhead applied to products at a rate of 150% of direct labor cost. Treating each case independently, find the...
-
Sunset Graphics is considering two mutually exclusive projects. Both require an initial investment of $100,000. Assume a marginal interest rate of 10 percent and no residual value for either...
-
How does each of the following affect the solubility of an ionic compound? (a) Lattice energy, (b) Solvent (polar versus nonpolar), (c) Enthalpies of hydration of cation and anion
-
The concentration of commercially available concentrated sulfuric acid is 98.0 percent by mass, or 18 M. Calculate the density and the molality of the solution.
-
The teachers of 8-year-old Jack are concerned that he has recently begun to withdraw socially and to show little interest in schoolwork.
-
Electromagnetic induction The purpose of this exercise is to determine the direction of the induced current in a circular loop by two different methods. Consider a circular conducting loop of radius...
-
Find the magnitude of the resultant force (FR) and the angle (p) it makes measured counterclockwise from the positive X-axis, given: FA= 50 lbs, 0A = 56, FB = 24 lbs, B = 28 FR = Number = Number lbs O
-
For the shapes below, what is the moment of force BC about axis OA? 0.925 m G 0.35 m 0.5 m B 0.875 m 0.5 m O (r(OD)XF(BH)).landa(OH) O landa(OD).(r(OB)XF(BH)) O F(OA).(landa(AD)Xr(AB)) O landa (OA)....
-
Q3: A mass is oscillated on a spring with the function as x= 6 sin 5T t where x is in metre and t is in seconds. i. Sketch the graph of displacement against time. ii. Write the expression for the...
-
A particle with a mass M = 68.4 MeV/c at rest decays into two particles. The first with mass m = 7.2 MeV/c and the second one is massless (i.e. a photon). It is given that the life-time of the...
-
The balance of the Estimated Warranty Liability account was $25,000 on January 1, 2022, and $34,400 on December 31, 2022. Based on an analysis of warranty claims during the past several years, this...
-
Q1) What is the a3 Value Q2) What is the a7 Value Q3) What is the a4 Value Q4) What is the b3 Value Q5) What is the b2 Value Q6) What is the sign of 2nd constraint? A pastry chef at a bakery wants to...
-
A knowledge of molar absorptivities is particularly important in biochemistry, where UV spectroscopy can provide an extremely sensitive method of analysis. For example, imagine that you wanted to...
-
Which of the following compounds would you expect to show ultraviolet absorptions in the 200 to 400nmrange? (a) (b) (c) CN CH3 (f) (d) (e) " N. Indole Aspirin
-
Show the structures of all possible adducts of the following diene with 1 equivalent ofHC1:
-
Rowland & Sons Air Transport Service, Incorporated, has been in operation for three years. The following transactions occurred in February: February 1 Paid $ 5 5 5 to rent equipment that was used and...
-
Comparative Statement of Financial Position for 2 0 X 4 and 2 0 X 3 , a Statement of income for 2 0 X 4 , and additional information from the accounting records of Olaniyi Industries, are provided...
-
When Patey Pontoons issued 6 % bonds on January 1 , 2 0 2 4 , with a face amount of $ 6 0 0 , 0 0 0 , the market yield for bonds of similar risk and maturity was 7 % . The bonds mature December 3 1 ,...

Study smarter with the SolutionInn App


