Which of the following compounds would you expect to show ultraviolet absorptions in the 200 to 400nmrange?
Question:
Which of the following compounds would you expect to show ultraviolet absorptions in the 200 to 400nmrange?
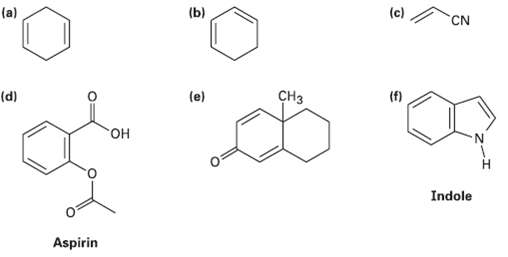
Transcribed Image Text:
(a) (b) (c) CN CH3 (f) (d) (e) "он N. Indole Aspirin
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (15 reviews)
All compounds having alternating single and mu...View the full answer

Answered By

Benish Ahmad
I'm a professional software engineer. I'm lectutrer at GCUF and I have 3 years of teaching experience. I'm looking forward to getting mostly computer science work including:
Programming fundamentals
Object oriented programming
Data structures
object oriented design and analysis
Database system
Computer networks
Discrete mathematics
Web application
I am expert in different computer languages such as C++, java, JavaScript, Sql, CSS, Python and C#. I'm also have excellent knowledge of essay writing and research. I have worked in other Freelancing website such as Fiverr and Upwork. Now I have finally decided to join the SolutionInn platform to continue with my explicit work of helping dear clients and students to achieve their academic dreams. I deliver plagiarism free work and exceptional projects on time. I am capable of working under high pressure.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Related Video
For this experiment, we\'ve compared the freshness of flowers by keeping them in three separate bottles filled with water, aspiring water, and food plant
Students also viewed these Organic Chemistry questions
-
Which of the following compounds would you expect to be the most generally reactive, and why? DO
-
Which of the following compounds would you expect to be most acidic? Justify your choice.
-
Which of the following compounds would you expect to exhibit only London forces? a. Potassium chloride, KCl b. c. Silicon tetrafluoride, SiF4 d. Phosphorus pentachloride, PCl5 (
-
Cost information for Lake County Library is as follows. In addition to directly traceable costs, the library incurred $24,000 for a building lease. REQUIRED A. Allocate to departments any costs that...
-
What are some of the challenges with intercultural and cross-cultural communications and leadership?
-
Each of the three molecules shown here contains an OH group, but one molecule acts as a base, one as an acid, and the third is neither acid nor base. (a) Which one acts as a base? Why does only this...
-
Within the chapter, three alternative statistical sampling plans are introduced and illustrated for use when conducting tests of controls under the second standard of field work: attribute estimation...
-
SallyMay, Inc., designs and manufactures T-shirts. It sells its T-shirts to brand-name clothes retailers in lots of one dozen. SallyMays May 2013 static budget and actual results for direct inputs...
-
You are studying the reproductive success of a species of smallcoral reef fish, Lacustrina didactus , on Dariada Reef inAustralia. This species reproduces by spawning. That is, a male andfemale come...
-
write a closed-research legal memo using the case cases given, addressing whether Mr. Adler can assert an adverse possession claim to Scrub Lot 40. Please write up your memo using the issue , brief...
-
A knowledge of molar absorptivities is particularly important in biochemistry, where UV spectroscopy can provide an extremely sensitive method of analysis. For example, imagine that you wanted to...
-
Show the structures of all possible adducts of the following diene with 1 equivalent ofHC1:
-
Use the various properties of exponential and logarithmic functions to evaluate the expressions in parts (a) (c). Given (x) = 3 x , find (c) 8(In). (b) g(ln 5) (a) f(log; 2) (b) f(log3 (In 3)) (c)...
-
1. Nail & Hammer Shack purchased 246 table fans of a particular make. The unit cost was $29.50 and the regular selling price was $60.00 each. The store sold 122 of them at the regular selling price....
-
Abby is single and age 25. She has AGI of $110,000 and incurs the following expenditures in 2022: Medical expenses: $9,000 Interest on home mortgage (mortgage was $300,000): $11,000 State income tax:...
-
Ms. Leslie Little is a full time architect. To help finance her various hobbies, she grows carrots for sale in a little plot next to her house. Although she has no farming background, in most years...
-
Susanne and David are married and not self-employed. Susanne pays $7,180.09 in Social Security tax, and contributes $22,500 to a 401(k) plan through her employer. David pays $2,204.55 in Medicare tax...
-
Alpine Ltd signed an 8-year non-cancellable lease with Mt Buller Ltd for the use of high-tech equipment. The following information is available: Lease term 8 years Salvage value at the end of the...
-
Three forces are exerted on a \(2.00-\mathrm{kg}\) block initially at rest on a slippery surface: a \(100-\mathrm{N}\) force directed along the positive \(x\) axis, a \(50.0-\mathrm{N}\) force that...
-
Medi-Exam Health Services, Inc. (MEHS), located in a major metropolitan area, provides annual physical screening examinations, including a routine physical, EKG, and blood and urine tests. MEUS's...
-
What are the common units for expressing solution concentration?
-
Propose a mechanism that shows why p-chlorotoluene reacts with sodium hydroxide at 350 C to give a mixture of p-cresol and m-cresol.
-
Propose mechanisms and show the expected products of the following reactions. (a) 2, 4-dinitrochlorobenzene + sodium methoxide (NaOCH3) (b) 2, 4-dimethylchlorobenzene + sodium hydroxide, 350 oC (c)...
-
Nucleophilic aromatic substitution provides one of the common methods for making phenols. (Another method is discussed in Section 19-17.) Show how you would synthesize the following phenols, using...
-
Gold Star Rice, Limited, of Thailand exports Thai rice throughout Asia. The company grows three varieties of rice-White, Fragrant, and Loonzain. Budgeted sales by product and in total for the coming...
-
How much discount interest will there be if 8 2 , 5 0 0 is due at the end of 4 . 5 years with 1 0 . 9 % interest rate
-
Briefly explain the role of organizational culture in organization design. Classify IKEA'S organization's culture and Explain how you arrived at the classification. If the current culture at IKEA...

Study smarter with the SolutionInn App


