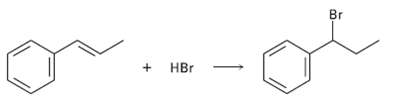
Addition of HBr to 1-phenyipropene yields only (1-bromopropyl) benzene. Propose a mechanism for the reaction, and explain
Question:
Addition of HBr to 1-phenyipropene yields only (1-bromopropyl) benzene. Propose a mechanism for the reaction, and explain why none of the other regioisomer isproduced.
Transcribed Image Text:
Br + HBr
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 53% (13 reviews)
HBr o Protonation of the double bond ...View the full answer

Answered By

Joseph Mwaura
I have been teaching college students in various subjects for 9 years now. Besides, I have been tutoring online with several tutoring companies from 2010 to date. The 9 years of experience as a tutor has enabled me to develop multiple tutoring skills and see thousands of students excel in their education and in life after school which gives me much pleasure. I have assisted students in essay writing and in doing academic research and this has helped me be well versed with the various writing styles such as APA, MLA, Chicago/ Turabian, Harvard. I am always ready to handle work at any hour and in any way as students specify. In my tutoring journey, excellence has always been my guiding standard.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Propose a mechanism for the reaction of acetyl chloride with phenylmagnesium bromide to give 1,1-diphenylethanol. OH (1) ether solvent CH3-C CI 2 (2) H 0 acetyl chloride phenylmagnesium bromide...
-
Propose a mechanism for the reaction of cyclohexyl methyl ketone with excess bromine in the presence of sodium hydroxide.
-
Propose a mechanism for the reaction of an amide with thionyl chloride to form a nitrile.
-
How did your parents communication with you influence your self-concept?
-
Sam, age 45, is single. For 2016, he has the following items: Business income........................................................$70,000 Business...
-
What do college students do with their time? A survey of 3,000 traditional-age students was taken, with the results as follows: ActivityPercentage (%) Attending class/lab........................ 9...
-
The following situations were not discovered in the audit of Pars Company by an inexperienced staff assistant: 1. Several accounts were incorrectly aged on the client's aging schedule. 2. Interest...
-
The post-closing trial balance of Anderson Company at December 31, 2011, is shown here. During 2012, Anderson Company had the following transactions: a. Inventory purchases were $80,000, all on...
-
What are the most common stress-related disorders, and what innovative treatments are currently being developed to address these conditions ?
-
The Motion Picture Association of America studies the ethnicity of moviegoers to understand changes in the demographics of moviegoers over time. Here are the numbers of moviegoers (in millions)...
-
Electrophilic substitution on 3-phenylpropanenitrile occurs at the ortho and Para positions, but reaction with 3-phenylpropenenitrile occurs at the meta position. Explain using resonance structures...
-
Triphenylmethane can be prepared by reaction of benzene and chloroform in the presence of A1Cl3. Propose a mechanism for thereaction. H. + CHCI3 AICI3
-
Parent Company holds bonds of its subsidiary, Sub Company, that were purchased on the open market at a substantial discount. Upon consolidation of Parents financial statements, how should the...
-
For the month of May, the balance in Acme Company's raw materials inventory account was $20,500 at the beginning of the month and $37,000 at the end of the month. Raw materials purchases during the...
-
A company has the following adjusted trial balance: Debit Credit Cash Accounts receivable Prepaid rent Equipment 1,500 2,100 100 3,500 Accumulated depreciation-Equipment 1,500 Accounts payable...
-
The business determines that the interest expense on a note payable for the period ending December 31 is $490. This amount is payable on January 1. Journalize the entries required on December 31 and...
-
Stacey's Plano Rebuilding Company has been operating for one year. At the start of the second year, its Income statement accounts had zero balances and its balance sheet account balances were as...
-
Why do we typically consider economies where the endowment when young is greater than the endowment when old? Consider a model economy as described in Module 1, but assume that workers are endowed...
-
How does tax avoidance differ from tax deferral? Explain whether either of these is a form of tax evasion.
-
(a) Given a mean free path = 0.4 nm and a mean speed vav = 1.17 105 m/s for the current flow in copper at a temperature of 300 K, calculate the classical value for the resistivity of copper. (b)...
-
Derive the following Maxwell relations for open systems. a. Starting from Eq. 6.2-5a, b. Starting from Eq. 6.2-6a, c. Starting from Eq. 6.2-7a, d. Starting from Eq. 6.2-8a, (). HT ON (0) aN S, N S,V...
-
Show the products of thesereactions: CH NaOH Br2 Bra b) a) . .
-
The reaction of an alkenes with bromine in an alcohol as solvent produces as ether as the product. Show a mechanism for the following reaction and explain the stereochemistry of theproduct. Br . H....
-
Show all the steps in the mechanism for the formation of MTBE from methanol and isobutylene.
-
Privack Corporation has a standard cost system in which it applies overhead to products based on the standard direct labor-hours allowed for the actual output of the period. Data concerning the most...
-
HomeLife Life Insurance Company has two service departments ( actuarial and premium rating ) and two production departments ( advertising and sales ) . The distribution of each service department s...
-
Otsego Industries manufactured 300,000 units of product last year and identified the following costs associated with the manufacturing activity: Variable costs: Direct materials used Direct labor...

Study smarter with the SolutionInn App


