Triphenylmethane can be prepared by reaction of benzene and chloroform in the presence of A1Cl3. Propose a
Question:
Triphenylmethane can be prepared by reaction of benzene and chloroform in the presence of A1Cl3. Propose a mechanism for thereaction.
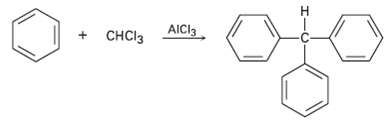
Transcribed Image Text:
H. + CHCI3 AICI3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (10 reviews)
CHCl3 AICI3 CHC CHCI2 CHCI AICI4 CHC1 CIAI Cl3 D...View the full answer

Answered By

Sumit kumar
Education details:
QUATERNARY Pursuing M.Tech.(2017-2019) in Electronics and Communication Engg. (VLSI DESIGN) from
GNIOT Greater Noida
TERTIARY B.Tech. (2012-2016) in Electronics and Communication Engg. from GLBITM Greater Noida
SECONDARY Senior Secondary School Examination (Class XII) in 2012 from R.S.S.Inter College, Noida
ELEMENTARY Secondary School Examination (Class X) in 2010 from New R.J.C. Public School ,Noida
CERTIFICATION
Summer Training in ‘WIRELESS EMBEDDED SYSTEM’ from ‘XIONEE’ for the six weeks.
EMBEDDED SYSTEM Certificate issued by CETPA INFOTECH for one day workshop.
Certificate of Faculty development program on OPTICAL COMMUNICATION and NETWORKS for one week.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The following alkyl bromide can be prepared by reaction of the alcohol (S)-2-pentanol with PBr3. Name the compound, assign (R) or (S) stereochemistry, and tell whether the reaction of the alcohol...
-
Ethers can be prepared by reaction of an alkoxide or phenoxide ion with a primary alkyl halide. Anisole, for instance, results from reaction of sodium phenoxide with iodomethane. What kind of...
-
Norbornadiene (Problem 14.41) can be prepared by reaction of chloroethylene with 1, 3-cyclopentadiene, followed by treatment of the product with sodium ethoxide. Write the overall scheme, and...
-
How would you compare the value of direct definition from friends in face-to-face conversation and strangers comments on a YouTube posting?
-
The $1 million maximum compensation deduction does not seem to have deterred large corporations from remunerating their executives at very high levels. What techniques are being used to work around...
-
If a 15-cm-long column has a plate height of 5.0 m, what will be the half-width (in seconds) of a peak eluted at 10.0 min? If plate height = 25 m, what will be w1/2?
-
To the left of z = 2.56 and to the right of z = 1.25 Find the indicated area under the standard normal curve. If convenient, use technology to find the area.
-
The following journal entries are recorded in Kiera Co.s process cost accounting system. Kiera produces handbags and scarves. Overhead is applied to production based on direct labor cost for the...
-
Argue that international space law is adequate and will explain why, using examples. Argue that current space law is not adequate and explain why, using examples. Cite source
-
One of the major measures of the quality of service provided by an organization is the speed with which the organization responds to customer complaints. A large family-held department store selling...
-
Addition of HBr to 1-phenyipropene yields only (1-bromopropyl) benzene. Propose a mechanism for the reaction, and explain why none of the other regioisomer isproduced. Br + HBr
-
At what position, and on what ring, would you expect the following sub stances to undergo electrophilicsubstitution? (a) (b) CH3 Br (c) (d) .CI CH3 Z O=U
-
When patients suffered from hemorrhagic fever, M*A*S*H doctors replaced lost sodium by administering a saline solution intravenously. However, headquarters (HQ) sent a treatment change disallowing...
-
Compare and contrast a labor-based forward rate agreement (interest rate forward) to a euro dollar future. Discuss their respective dates, trading venue, settlement, and convexity.
-
What considerations (ethical or otherwise) do professional sports leagues prioritize when making decisions about relocating teams, particularly in light of the economic impact on local communities...
-
On March 1, Murray signed a contract to pay Anton $2000 plus interest at 8% on June 1, and $3000 plus interest at 8% on September 1. Anton sold the contract to Randy on May 1 at a price negotiated to...
-
WHAT ARE THE POSSSIBLE RICK FACTORS WHILE BUILDING A BUSSINESS ? Explain
-
Can a business-to-business marketer be innovative in the way a brand is built? What are requirements for ingredient brandings, and what are current examples? What industries are more inclined to use...
-
Based upon Deweys chosen investment process for the management of the Purity Fund, rebalancing of the fund will most likely occur: A. at regular intervals. B. in response to changes in...
-
Identify Thank You mission, strategy and core competencies. Identify strategy changes that have taken place at Thank You since its founding in 2008. Your answer must in text references and must be...
-
Redo Problem 5.27 using Aspen Plus. Problem 5.27 Methane at 260 K is to be isothermally compressed from 0.1 MPa to 1.0 MPa. a. What is the minimum work required, and how much heat must be removed to...
-
Show the products of thesereactions: 1) Hg(O,CCH,) . 2) NaBH3. NAOH 1) Hg(O,CCH,)2, H,0 2) NaBH4, NaOH b) a) . H SO4 . H2SO4 d) HgSO, H9SO,
-
Explain which of the reaction would provide a better synthesis of3-hexanone. . . CH,CH,CH,CH,CH, 3-Hexanone CH,CH,C=CCH CH3 CH,C=CCH,CH,CH3 H,SO4 HgSO, H&SO,
-
Show the products of thesereactions: 1) BH3, THF 1) BH3. THF 2) .. NaOH b) 2) ,, NaOH 1) BH3, THF 2) H,O2. NaOH CH-CH3
-
Consider the vectors u=2ij2k and v=2i+2j+4k. Find the vector components of u that are parallel and perpendicular to v.
-
1. Determine whether the following sets form subspaces and justify your answers. (a) {(1, 2): 122 = 0} in R (b) {(1, 2) |1|-|x2|} in R2 (c) {(x1, x2, x3): x3 = 21 or x3 = 12} in R3 (d) {(x1, x2, x3):...
-
Solve the system of two equations for y two equations are: A1=ax+by A2=cx+dy

Study smarter with the SolutionInn App


