All naturally occurring amino acids have the same relative configuration. All have the S absolute configuration, except
Question:
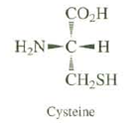
All naturally occurring amino acids have the same relative configuration. All have the S absolute configuration, except for cysteine, which has the R configuration.Explain.
Transcribed Image Text:
CO.H H,N-C-H CH,SH Cysteine
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 68% (16 reviews)
In all of the amino acids except cysteine the CO 2 H group has priority ...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Most naturally occurring amino acids have chirality centers (the asymmetric carbon atoms) that are named (S) by the Cahn-Ingold-Prelog convention (Section 5-3). The common naturally occurring form of...
-
The 20 naturally occurring amino acids (See the following table) are all l amino acids, and they all have the S configuration, with the exception of glycine (which lacks a chirality center) and...
-
Identify which 2 of the 20 naturally occurring amino acids are expected to have the same pI.
-
1. As a policy maker you should never worry much about those are eligible for Medicaid benefits and do not enroll. This is because they will enroll in public insurance if they need it. True or False?...
-
Classify the following organisations as professional service firms, mass service entities or service shops. In each case, explain the reasons for your classification. 1. Motor registration department...
-
Fredrick Wilson Company determined that one of its finite- life intangible assets is impaired. The assets net carrying value on the date of the impairment is $ 905,000. Fredrick Wilson does not use a...
-
Where are UCs documented?
-
The Russell Company has an extensive job-costing facility that uses a variety of metals. Consider each requirement independently. Required 1. Job 372 uses a particular metal alloy that is not used...
-
Cooper, a member of a forensics investigation team, was investigating a cyber-attack performed on an organization. During the investigation process, Cooper secured the incident area and collected all...
-
Stan Baiman owns a catering company that prepares food and beverages for banquets and parties. For a standard party the cost on a per-person basis is as follows: Food and beverages ........... $ 30...
-
Draw a stereo isomer of this compound that is chiral, and draw two that are not chiral? CH3 -
-
Explain whether each of these compounds is chiral or not: , CCH3 C=C=C] CH3 . a) b) C=C=C, , , . c) CH;O. `CH3 - . d) CH;O CO,H CH;OS Br CH;CH2 e) f) N-CH,CH3 Ph CH;CH2
-
In what financial activities does a corporate treasurer engage?
-
If r is the instantaneous position vector for a particle, show that the velocity and acceleration of the particle are given by (use Eq. A.7-2): d V= r = 8,7 +8,10 +8 dt a = 8,( r) + (r + 2) + 8 - in...
-
Using the Fe-C phase diagram, answer the following questions: (a) (5p.) For a 0,76%C steel, what is the amount of total ferrite at 400C? (b) (5p.) The weight percentages of proeutectoid ferrite and...
-
Given: k1= 80 N/mm, k2 = 100 N/mm, k3 = 160 N/mm, F3 = 200 N, F4 = 100 N, and nodes 1 and 2 are fix a. Global stiffness matrix b. Displacements of nodes 3 and 4 c. Reaction Forces at nodes 1 and 2 k...
-
Derive an expression for inverse of a second order tensor in index notation: dA- dA
-
Q1. For refrigerant 134a, determine the values specified for each of the following conditions: p = 140 lb/in p = 20 lb/in P = 200 psi P = u = 134.09 BTU/lb T = 75F v = 0.1865 ft/lb 150 psi T = BTU/lb...
-
How are flexible-budget and sales-volume variances calculated?
-
Suppose Green Network Energy needs to raise money to finance its new manufacturing facility, but their CFO does not think the company is financially capable of making the periodic interest payments...
-
Simplify each expression. 7+2(-10x + 4)
-
Ribose, an essential part of ribonucleic acid (RNA), has the following structure: (a) How many chirality centers does ribose have? Identify them. (b) How many stereo isomers of ribose are there? (c)...
-
On catalytic hydrogenation over a platinum catalyst, ribose (Problem 9.57) is converted into ribitol. Is ribitol optically active or inactive?Explain. CH2 Ribitol
-
Hydroxylation of cis-2-hutene with OsO4 yields 2, 3-butanediol. What stereochemistry do you expect for the product?
-
A horizontal board of negligible thickness and area 4.0 m hangs from a spring scale that reads 60 N when a 2.0 m/s wind moves below the board. The air above the board is stationary. When the wind...
-
A force in the positive direction of an x axis acts on an object moving along that axis. If the magnitude of the force is F = 7.2e*/3.1 N, with x in meters, find the work done by as the object moves...
-
The only force acting on a 2.2 kg body as it moves along the positive x axis has an x component Fx = -5x N, where x is in meters. The velocity of the body at x = 2.5 m is 9.0 m/s. (a) What is the...

Study smarter with the SolutionInn App


