Although 2-substituled 2-cyclopentenones are in a base-catalyzed equilibrium with their 5-substituted 2-cyclopentenone isomers (Problem 22.38), the analogous
Question:
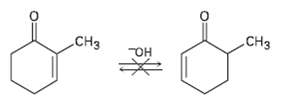
Although 2-substituled 2-cyclopentenones are in a base-catalyzed equilibrium with their 5-substituted 2-cyclopentenone isomers (Problem 22.38), the analogous isomerization is not observed for 2-substituted 2-cyclohexenones.Explain.
Transcribed Image Text:
.CHз .CHз он
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
Protons a to a carbonyl group or to an enone carbonyl group are acidic Problem 22...View the full answer

Answered By

Muhammad adeel
I am a professional Process/Mechanical engineer having a vast 7 years experience in process industry as well as in academic studies as a instructor. Also equipped with Nebosh IGC and lead auditor (certified).
Having worked at top notch engineering firms, i possess abilities such as designing process equipment, maintaining data sheets, working on projects, technical biddings, designing PFD and PID's etc.
Having worked as an instructor in different engineering institutes and have been involved in different engineering resrearch projects such as refinery equipment designing, thermodynamics, fluid dynamics, chemistry, rotary equipment etc
I can assure a good job within your budget and time deadline
4.90+
52+ Reviews
60+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Assume the numerals given are in a base 5 numeration system. Assume the numerals in this system and their equivalent HinduArabic numerals are Write the HinduArabic numerals equivalent to each of the...
-
Assume the numerals given are in a base 5 numeration system. Assume the numerals in this system and their equivalent HinduArabic numerals are Write the HinduArabic numerals equivalent to each of the...
-
Assume the numerals given are in a base 5 numeration system. Assume the numerals in this system and their equivalent HinduArabic numerals are Write the HinduArabic numerals equivalent to each of the...
-
A 0.1 cm thick flat copper plate, 2.5 m x 2.5 m square is to be cooled in a vertical position. The initial temperature of the plate is 90?C with the ambient fluid at 30?C. The fluid medium is either...
-
How can you ensure that you close a bad-news message pleasantly and maintain the reader's goodwill?
-
A mining company operates two mines, each producing three grades of ore. The West Summit mine can produce 2 tons of low-grade ore, 3 tons of medium-grade ore, and 1 ton of high-grade ore in one hour...
-
Explain how a system can learn by building decision trees, using the ID3 algorithm.
-
Seaweed Salvagers is a corporation engaged in producing foodstuffs from seaweed. Seaweeds primary salvaging plant is located in California. Because salvaging technology hasnt changed through the...
-
a) Is the code below tightly coupled if yes provide a reason and implement a loosely coupled working version of it? (7 marks) public class classVolume { public static void main(String args[]) { Box b...
-
Claire Consultants has been entrusted with the task of evaluating business plan that has been divided into four sectionsmarketing, finance, operations and human resources. Chris, Steve, Juana, and...
-
An interesting consequence of the base-catalyzed isomerization of unsaturated ketones described in Problem 22.37 is that 2-substituted 2-cyclo- pentenones can be inter converted with 5-substituted...
-
Using curved arrows, propose a mechanism for the following reaction, one of the steps in the metabolism of the amino acidalanine. 2-02 -032 CH H Co co2 Base . CH - CH
-
Verify the conclusion of Greens Theorem by evaluating both sides of Equations (3) and (4) for the field F = Mi + Nj. Take the domains of integration in each case to be the disk R: x 2 + y 2 a 2 and...
-
Harvison Corporation has bonds outstanding with a face value of $1,000. The required rate of return on these bonds is currently 12%, and interest is paid semiannually. The bonds mature in 7 years,...
-
Decide on a start-up business you would like to get off the ground. shows what product or service the business will provide, how they will get funding to get the business up and running, and what...
-
2. Convert binary 1110101100012 to decimal, hexadecimal and octal. 3. Convert Hexadecimal 8FC216 in decimal, binary and octal.
-
Using the law of accumulation and the law of population, explain how the division of labor might contribute to the concentration of wealth in the hands of a few.
-
What types of nonpublic agency records would each agency keep that would be of interest to a criminaljustice researcher? Give an example of a research question that could be answered by access to the...
-
Show that liquid fresh water makes up about 0.50, of the water on Earth based on the data in Figure 24.18. Figure 24.18 Salt water in oceans: 97.6% Ice caps and glaciers: 1.9% Groundwater: 0.49%...
-
Calculate the electrical conductivity of a fiber-reinforced polyethylene part that is reinforced with 20 vol % of continuous, aligned nickel fibers.
-
Imagine that a molecule with six electron groups is confined to two dimensions and therefore has a hexagonal planar electron geometry. If two of the six groups are lone pairs, where are they located?...
-
Complete the reactions given in Fig. P19.45 by giving the principal organic product(s). Fig. P19.45 (a) (b) (c) p-toluenesulfonic acid (catalyst CH, t CH,OH - (solvent) ether Hio -caphenone, +...
-
Using known reactions and mechanisms discussed in the text, complete the reactions given in Fig. P19.46 on p. 942. Fig. P19.46 NaBH4 CH OH
-
A compound A, C8H8O, when treated with Zn amalgam and HCl, gives a xylene (dimethylbenzene) isomer that in turn gives only one ring monobromination product with Br2 and Fe. Propose a structure for A.
-
The article "SAC Case Tests a Classic Dilemma" describes how certain aspects of the government's insider trading case against SAC Capital resembles the standard prisoner's dilemma game as well as...
-
Lamar's capital balance is $40,960 after admitting Terrell to the partnership by investment. If Lamar's ownership interest is 20% of total partnership capital, what were (1) Terrell's cash investment...
-
+ The cash balances per books for the Jackson Company on November 30, 2002 and December 31, 2003, are $3,625 and $10,967 respectively. The following checks and receipts were recorded on the books for...

Study smarter with the SolutionInn App


