Although carbon?carbon double bonds are shorter than carbon?carbon single bonds, all of the carbon?carbon bonds of benzene
Question:
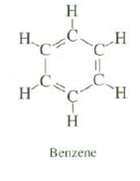
Although carbon?carbon double bonds are shorter than carbon?carbon single bonds, all of the carbon?carbon bonds of benzene are the same length. Explain.
Transcribed Image Text:
H. H C. H H Benzene H H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
The actual structure of benzene is a h...View the full answer

Answered By

Stacy kosgei
I offer quality, original and timely services; Highly credible and void of plagiarism. Your success is my pleasure.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The hydrocarbons acetylene (C2H2) and benzene (C6H6) have the same empirical formula. Benzene is an "aromatic" hydrocarbon, one that is unusually stable because of its structure. (a) By using the...
-
Bonds between carbon and oxygen (C O) are more polar than bonds between sulfur and oxygen (S O). Nevertheless, sulfur dioxide (SO 2 ) exhibits a dipole moment while carbon dioxide (CO 2 ) does not....
-
Although perpetual bonds are illegal in the United States, sometimes it is easiest to assume that interest payments last forever to show some simple results based on Equation. Use that equation to...
-
If we take $1 away from a rich person and give it to a poor person, the rich person loses less utility than the poor person gains. Comment.
-
Visit the website of a major airline to find information about the airline's mission, objectives, strategic priorities, organizational structure and operations. Required: 1. Outline the airline's...
-
A firm has a generous but rather complicated policy concerning end-of-year bonuses for its lower-level managerial personnel. The policys key factor is a subjective judgment of contribution to...
-
Explain the idea behind the following heuristics: most-constrained variable most-constraining variable least-constraining variable min-conflicts
-
Mary is considering opening a new grocery store in town. She is evaluating three sites: downtown, the mall, and out at the busy traffic circle. Mary calculated the value of successful stores at these...
-
What is a 3-way match? Part 2: Why is the 3-way match considered an internal control? Part 3: Who is responsible for performing the 3-way match, the Purchasing department, the Receiving department,...
-
You are evaluating various investment opportunities currently available and you have calculated expected returns and standard deviations for five different well-diversified portfolios of risky...
-
Chlorine is more electronegative than phosphorus. Predict the dipole moment of PCl5.
-
Explain whether or not these molecules are polar (have a dipole moment). (a) CBr 4 (b) NH 3 (c) CH 3 OCH 3 (d) CH 2 C1 2 (e) CO 2
-
Briefly describe what an investment timing option is and why such options are valuable.
-
46) When you are analyzing mutually exclusive projects, if you have a conflict between NPV and IRR, you are better off to make the final choice as to which project to choose 44) Find the...
-
1. What is one of the drinks that help you remember the debit rules? Choose One. 20 points Debit ADE Mohito GIN and Tonic Lemonaade 2. What is one of the drinks that help you remember the credit...
-
Assume that calendar year 2017 ended a few weeks ago. Prepare Soaring Companys Balance Sheet at December 31, 2017 and its Income and Expense Statement for calendar year 2017 from the information in...
-
maturity years to maturity ask price calculated yield 5/15/2013 1 99.883 0.001171371 5/15/2014 2 99.387 0.003079164 5/15/2015 3 98.606 0.004690323 5/15/2016 4 97.163 0.007220998 5/15/2017 5 95.412...
-
Assume a Hotelling line of a distance l = 1. There are two companies A and B, both of them being located at a longer distance. Company A is placed closer to the end of the line, while company B is...
-
Explain the difference between an emissions trading scheme and a carbon tax. What are some of the benefits and limitations of each?
-
CRUZ, INC. Comparative Balance Sheets December 31, 2015 CRUZ, INC. Income Statement For Year Ended December 31, 2015 Required Use the indirect method to prepare the cash provided or used from...
-
Determine an appropriate viewing rectangle for the function f(x) = 8 2x 2 and use it to graph f.
-
How many products might be formed on chlorination of o-xylene (o-dimethyl-benzene), m-xylene, and p-xylene?
-
When benzene is treated with D2SO4, deuterium slowly replaces all six hydrogens in the aromatic ring. Explain.
-
Which of the following alkyl halides would you expect to undergo Friedel-Crafts reaction without rearrangement? Explain. (a) CH3CH2Cl (b) CH3CH2CH (Cl) CH3 (c) CH3CH2CH2Cl (d) (CH3) CCH2Cl (e)...
-
Suppose you bought a new home for $210,000 using a 30-year mortgage with monthly payments of $1,218.841. The annual interest rate of the mortgage is 5.7%. After the first 3 years (36 monthly...
-
Rather than use the hypothetical information above, determine from available news sources if the Fed is increasing, holding steady, or decreasing interest rates. Similarly, is the Federal Government...
-
For a two-sided matching problem, we say that matching is Pareto efficient, if there is no matching ', such that and '(x) R (x) for all x WUF, '(x) Px (x) for some x WUF. Show that stability implies...

Study smarter with the SolutionInn App


