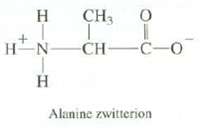
Amino acids such as alanine actually exist as species called zwitterions, with a positive charge on the
Question:
Amino acids such as alanine actually exist as species called zwitterions, with a positive charge on the nitrogen and a negative charge on the oxygen. Explain what effect you expect this to have on the melting point of alanine.
Transcribed Image Text:
CH3 0 T HN CH- HINIH Н Н I -C-0 Alanine zwitterion
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (17 reviews)
The zwitterions form of al...View the full answer

Answered By

Somshukla Chakraborty
I have a teaching experience of more than 4 years by now in diverse subjects like History,Geography,Political Science,Sociology,Business Enterprise,Economics,Environmental Management etc.I teach students from classes 9-12 and undergraduate students.I boards I handle are IB,IGCSE, state boards,ICSE, CBSE.I am passionate about teaching.Full satisfaction of the students is my main goal.
I have completed my graduation and master's in history from Jadavpur University Kolkata,India in 2012 and I have completed my B.Ed from the same University in 2013. I have taught in a reputed school of Kolkata (subjects-History,Geography,Civics,Political Science) from 2014-2016.I worked as a guest lecturer of history in a college of Kolkata for 2 years teaching students of 1st ,2nd and 3rd year. I taught Ancient and Modern Indian history there.I have taught in another school in Mohali,Punjab teaching students from classes 9-12.Presently I am working as an online tutor with concept tutors,Bangalore,India(Carve Niche Pvt.Ltd.) for the last 1year and also have been appointed as an online history tutor by Course Hero(California,U.S) and Vidyalai.com(Chennai,India).
4.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Why are amino acids such as arginine and histidine required in relatively large amounts by children but in smaller amounts by adults? The adult human is not able to make these amino acids.
-
Calculate the formal charge on the nitrogen atom in ammonia, NH3; in the ammonium ion, NH4+; and in the amide ion, NH2.
-
If you painted positive charge on the floor, what surface charge density would be necessary to suspend a 15C, 5.0-g particle above the floor?
-
Consider a soap bubble. Is the pressure inside the bubble higher or lower than the pressure outside?
-
The 'Peal life' suggests that senior executive salaries have risen considerably over time. What do Herzberg's theory of motivation and expectancy theory suggest about using large salary packages to...
-
Chad's Pottery Barn has enough clay to make 24 small vases or 6 large vases. He has only enough of a special glazing compound to glaze 16 of the small vases or 8 of the large vases. Let X1 = the...
-
To build an information system, which do you need to identify first, classes or objects?
-
Toddler Toys produces toy construction vehicles for young children. Plastic pieces are molded in the plastics department. These pieces are transferred to the assembly department, where direct...
-
Hi can you please answer this? Image transcription text Question 1 (a) (b) (C) (d) (Answcrallparts) (40 marks) (Compulsory) With respect to fatigue testing and analysis, explain what is meant by each...
-
The statement of financial position of Bharwani Traders on 31 October 204 is shown below. On 1 November 204 the following transactions took place: A cheque for $3 000 was paid to a credit supplier. A...
-
Glucose is a typical carbohydrate. What functional groups are present in glucose? What would you predict about the water solubility of glucose, oiler a reason why nature uses carbohydrates rather...
-
What is the DU of estrone, a female sex hormone? Use the number of carbons and oxygen?s in estrone to calculate the number of hydrogen?s it has. Estrone 0 CH3
-
Team A wins five out of six games in a series against team B. Find the maximum likelihood estimator of the probability that team A wins a game against team B. If you were willing to gamble, would it...
-
A jet engine burns a weak mixture \((\phi=0.32)\) of octane \(\left(\mathrm{C}_{8} \mathrm{H}_{18} ight)\) and air. The air enters the combustion chamber from the compressor at \(10 \mathrm{bar}\)...
-
A gas engine with a volumetric compression ratio of 10:1 is run on a weak mixture of methane \(\left(\mathrm{CH}_{4} ight)\) and air, with an equivalence ratio, \(\phi=0.9\). If the initial...
-
The following exercise helps clarify the difference between statements of fact and statements of inference. Place a familiar item (such as a coffee cup, a chair, or a ballpoint pen) in front of the...
-
If it is assumed that the enthalpy of reaction, \(Q_{p}\), is a constant, show that the value of \(K_{p}\) is given by \[K_{p}=\mathrm{e}^{-Q_{p} / \Re T+k}\] where \(k\) is a constant. For a...
-
Spend 10 minutes freewritingcontinuously, without judgmentabout a current assignment. How do you feel about the assignment and your work so far? What issues are you facing? What are your plans for...
-
Another problem with the open sites is that the students are posting personal information with the result that they are accessible by a nefarious element. Students' cell phone numbers, addresses,...
-
2.) Find the Laplace transform of f(t) 7e-St cos 2t +9 sinh2 2t. Use Laplace Table. %3D
-
Show that the average velocity of a car over a time interval [t 1 , t 2 ] is the same as the average of its velocities during the trip.
-
TMS ethers can be removed by treatment with fluoride ion as well as be acid-catalyzed hydrolysis. Propose a mechanism for the reaction of cyclohexyl TMS ether with LiF. Fluorotrimethyisilane is a...
-
Show the mechanism of the reaction of p-methaylphenol with 2-methylpropene and H3PO4 catalyst to yield the food additive BHT.
-
Assume that you need to prepare 5-cholesten-3-one from cholesterol. How could you use IR spectroscopy to tell whether the reaction was successful? What differences would you look for in the IR...
-
Explain how the economic conditions of Australia will affect the growth and planning of Telstra and optus carriers/service providers Evaluate how the political influences impact Telstra and optus...
-
Use the scores of 14 students on a history exam to answer problems 3-8. Find the 5-number-summary and draw a box plot of the scores. 54, 63, 65, 65, 70, 75, 77, 80, 82, 84, 88, 89, 95, 96. 3. What is...
-
Economic factors that can affect growth ? List five factors that affect economic growth and explain them. Political influences on public and commercial enterprises that provide services in networking...

Study smarter with the SolutionInn App


