What is the DU of estrone, a female sex hormone? Use the number of carbons and oxygen?s
Question:
What is the DU of estrone, a female sex hormone? Use the number of carbons and oxygen?s in estrone to calculate the number of hydrogen?s it has.
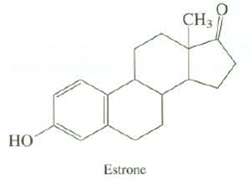
Transcribed Image Text:
НО Estrone 0 CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (6 reviews)
The Du of estrone is ...View the full answer

Answered By

Mahesh G
I have more than 7 years of experience in teaching physics, mathematics and python programming to more than 600 students including both online and offline tutoring.
I follow the following 7 step fundamental approach towards tutoring.
1. Curiosity, scope, enlightenment of the topic in hand.
2. Problem Definitions and elaboration.
3. Requisite mathematics, analytical abilities and quantitative
aptitude.
4. Preparing Algorithms for problem statement.
5. Concepts with analogies and building algorithm.
6. Introspection and improvising.
7. Daily class wise Cheat sheets(its not cheating) for consolidation.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
What is the Du Pont method for calculating return on investment? What is the advantage of this method?
-
What is the Du Pont Equation? And why is it useful?
-
The sex hormone estrone has been synthesized by a route that involves the following step. Identify the pericyclic reactions involved, and propose amechanism. CH CH Heat |CH30 CH0 Estrone methyl ether
-
Sketch the six graphs of the x- and y-components of position, velocity, and acceleration versus time for projectile motion with x 0 = y 0 = 0 and 0 < 0 < 90.
-
Outline the differences between extrinsic and intrinsic motivation, and explain how organisations can encourage intrinsic motivation.
-
Suppose that the average waiting time for a patient at a physicians office is just over 29 minutes. To address the issue of long patient wait times, some physicians offices are using wait-tracking...
-
Betsy is interested in predicting how many 75 -year-olds will develop Alzheimer's disease and is using as predictors level of education and general physical health graded on a scale from 1 to 10 ....
-
Refer to Problem 12-1. Return to the assumption that the company had $3 million in assets at the end of 2010, but now assume that the company pays no dividends. Under these assumptions, what would be...
-
You are a cybersecurity analyst with a few years of experience. A junior analyst just started their first role in cybersecurity at your company. Explain multifactor authentication (MFA) to the...
-
Complete an SFAS Matrix and a TOWS Matrix on your Strategic Audit firm using your EFAS Start with your EFAS and IFAS assignments. O Make all corrections to these assignments. The SFAS matrix is to...
-
Amino acids such as alanine actually exist as species called zwitterions, with a positive charge on the nitrogen and a negative charge on the oxygen. Explain what effect you expect this to have on...
-
Explain whether each pair of models represents isomers or the same compound. (All represent compounds with the formula C7H16.) Draw structures for each compound represented by the models.
-
A random sample of 10 students was taken from an introductory statistics class. The following data were obtained, where x denotes height, in inches, and y denotes score on the final exam. a. What...
-
1. Minal Enterprise invest RM 10000 now, RM5000 two years from now and RM 3000 five years from now. a) Determine how much money is accumulate at the end of seven years. b) Determine how much money...
-
An investment makes annual payments. The first payment of $608.00 is due in one year at t=1. Payments grow at a rate of 14.00% annually until t=22. Payments then are stable until t=33. Afterwards,...
-
For an investment, which will bring you 4 million TL in four years, how much should you pay today if inflation rate is 10% and interest without inflation is 5 % ? You can use the formula below where...
-
Mr. Vignesh Menan owns land (not ecologically sensitive land) with an ACB of $100,000 and a FMV of $170,000. In 20X2, he intends to donate this land to a registered Canadian charity and would like to...
-
Duracell is well known for manufacturing and marketing batteries. Recently they launched the Duracell Powermat, which is a wireless charger for mobile devices. Which brand development strategy did...
-
A 60-g Mars bar will supply you with \(1095 \mathrm{~kJ}\) of energy. How many of them would you need to get enough energy to climb the first \(800 \mathrm{~m}\) of the highest structure in the...
-
Fahrad Inc. sells all of its product on account. Fahrad has the following accounts receivable payment experience: Percent paid in the month of sale .........10 Percent paid in the month after the...
-
When a particle is located a distance x feet from the origin, a force of x 2 + 2x pounds acts on it. How much work is done in moving it from x = 1 to x = 3?
-
When the 1HNMR spectrum of an alcohol is run in dimethyl sulfoxide (DMSO) solvent rather than in chloroform, exchange of the OH proton is slow and spinspin splitting is seen between the OH proton...
-
Give IUPAC names for the following compounds: (a) (b) (c) (d)
-
Draw the structure of the carbonyl compound(s) from which each of the following alcohols might have been prepared, and show the products you would obtain by treatment of each alcohol with (i) Na...
-
Whirly Corporation's contribution format income statement for the most recent month is shown below: Sales (7,500 units) Variable expenses Contribution margin Fixed expenses Net operating income....
-
After she stopped at the stop sign and looked both ways, Donna pulled onto a roadway and turned right onto Johnson Road.She did not see the Plaintiff's vehicle traveling on Johnson Road. When the...
-
Fuji Co. is growing quickly. Dividends are expected to grow at a rate of 28 percent for the next three years, with the growth rate falling off to a constant 7 percent thereafter. If the required...

Study smarter with the SolutionInn App


