Among the most common over-the-counter drugs you might find in a medicine cabinet are mild pain relievers
Question:
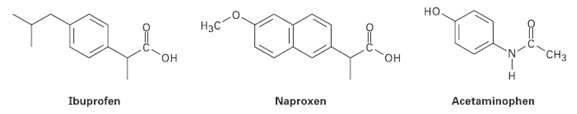
Among the most common over-the-counter drugs you might find in a medicine cabinet are mild pain relievers such ibuprofen (Advil, Motrin), naproxen (Aleve), and acetaminophen (Tylenol).
(a) How many sp3-hybridized carbons does each molecule have?
(b) How many sp2-hybridized carbons does each molecule have?
(c) Can you spot any similarities in theirstructures?
Transcribed Image Text:
но. Нас "CHз N. он он н Naproxen Acetaminophen Ibuprofen
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (16 reviews)
Ya HO Ibuprofen a b Acetaminophen Compound Ibuprofen Naproxe...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Draw a hierarchy for the components you might find in a graphical user interface. Note that some components can trigger actions. Some components may have graphics associated with them. Some...
-
Because merchants are in many places among the most common type of business, and because there are special issues in accounting for merchandising operations, this subject merits special...
-
Describe the type of inventory policy you might find in each of the following operations, and describe why a hospital, a cafeteria, an automobile repair facility, a bakery, and a dental office.
-
R. Bright, an electrical goods wholesaler, has three departments: (a) Music, (b) TV and (c) Kitchen. The following is a summary of Brights sales invoices during the week 1 to 7 February 2013. (a)...
-
Complete the following statements regarding special recapture provisions. a. Corporations selling depreciable real property are required to recapture as ordinary income the _________________ of two...
-
A solution contains 0.010 M Ba2+ and 0.010 M Ag+. Can 99.90% of either ion be precipitated by chromate (CrO42-) without precipitating the other metal ion?
-
An air heater in a large coal-fired steam generator heats fresh air entering the steam generator by cooling flue gas leaving the steam generator. One million \(\mathrm{lbm} / \mathrm{hr}\) of air at...
-
Eve Herschel is the owner of Herschels Pizza. Herschels is operated strictly on a carryout basis. Customers pick up their orders at a counter where a clerk exchanges the pizza for cash. While at the...
-
Kevin is a vice president of wealth management at a large branch of a financial services firm. In addition to managing a number of clients of his own, Kevin is the branch manager as well as the...
-
Melodic Musical Sales, Inc. is located at 5500 Fourth Avenue, City, and ST 98765. The corporation uses the calendar year and accrual basis for both book and tax purposes. It is engaged in the sale of...
-
There are two different substances that contain a carbon-carbon double bond and have the formula C4H8. Draw them, and tell how they differ.
-
Which element in each of the following pairs is more electronegative? (a) Li or H (b) B or Br (c) C1 or I (d) C or h
-
Laverne purchased a new piece of equipment to be used in its new facility. The $370,000 piece of equipment was purchased with a $50,000 down payment and with cash received through the issuance of a...
-
What policies are currently in place to address wealth and housing inequality in Canada? In your opinion, are these policies enough?
-
What role do social movements and collective action play in driving processes of social change, and how do theories of collective behavior, resource mobilization, and political opportunity structures...
-
How many years will it take Johnny to earn $1,122.00 if he invested $856.00 at 7% return?
-
Entrepreneurs should be aware of the following issues when trying to raise capital except: Will HR be outsourced or a manger hired to run the HR department internally? Will I get anything else...
-
What was CMS's conclusion regarding the practice restriction in AO- 2011-01? What factors did CMS consider in reaching this conclusion?
-
Which of the following types of style analysis use(s) a bottom-up approach to estimate the risk exposures in a portfolio? A. Returns-based style analysis only B. Holdings-based style analysis only C....
-
g(x) = x 5 5x 6 a. Show that g(x) = 0 has a root, , between x = 1 and x = 2. b. Show that the equation g(x) = 0 can be written as x = (px + q) 1/r , where p, q and r are integers to be found. The...
-
One question that arises in phase equilibrium calculations and experiments is how many phases can be in equilibrium simultaneously, since this determines how many phases one should search for....
-
Show the most stable conjugate base of these compounds: a) CHCHOH i b) HOCCHCHOH c) HNCHCHOH
-
Which of these species can behave as a Lewis acid? CI a) Cl-B CI H b) H-C-H H + c) CHCH
-
Which of these species can behave as a Lewis base? a) CH0CH,CH, CH3 d) CH3NH + b) CH3CHCH3 0: I.. e) CHCOH c) CH3NH
-
The setting of a piece of literature may be an important factor that influences the actions of the characters. Two literary works in which the setting plays an important role are The Story of an Hour...
-
Water is flowing in the pipe shown in the figure below, with the 8.45-cm diameter at point 1 tapering to 3.60 cm at point 2, located y = 13.0 cm below point 1. (a) point 1 m/s (b) point 2 m/s
-
A certain target is bombarded by electrons in an x-ray tube. The K, L, and M energy levels have energies 84.7, 14.3, and 2.97 keV, respectively. (a) What is the minimum value of the accelerating...

Study smarter with the SolutionInn App


