Which of these species can behave as a Lewis acid? CI a) Cl-B CI H b) H-C-H
Question:
Which of these species can behave as a Lewis acid?
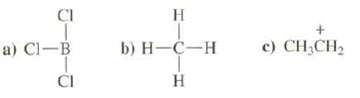
Transcribed Image Text:
CI a) Cl-B CI H b) H-C-H H + c) CH₂CH₂
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (12 reviews)
According to the Lewis acidbase definition an acid ...View the full answer

Answered By

Anoop V
I have five years of experience in teaching and I have National Eligibility in teaching (UGC-NET) .
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Which of these species can behave as a Lewis base? a) CH0CH,CH, CH3 d) CH3NH + b) CH3CHCH3 0: I.. e) CHCOH c) CH3NH
-
Indicate whether each of these species can act as an acid, a base, or both: H +1 a) H-N-H 1 H H e) H-C-O-H H b) H-O-H H: H TI f) H-C-C-C-H H H c) H-C-H H H d):Ci : g) H-O-C-:
-
Which compound is behaving as the Lewis acid and which as the Lewis base in this reaction? AICI3 T CHCHCHCH3 + AICI CHCHCHCH3
-
In an organization, managers communicate information downward to their departments and teams, and employees communicate information upward to their managers. If all members of an organization are not...
-
Explain the type of environment in a service organization that best suits the use of standard costs for control?
-
For a newborn full-term infant, the weight appropriate for gestational age is assumed to be normally distributed with = 3025 grams and = 165 grams. Compute the probability that a random sample of...
-
How is the SE Process Model applied to these development process models?
-
Anders Clothing manufactures embroidered jackets. The company uses a standard cost system to control manufacturing costs. The following data represent the standard unit cost of a jacket: Fixed...
-
1. What is the difference between an allowance and a reim-bursement? How does this difference affect labor burden? 2. Why are the social security and Medicare taxes paid by the employee not included...
-
The following exercises use the Purchase Orders database. Use MATCH and/ or INDEX functions to find the following: a. The row numbers corresponding to the first and last instance of item number 1369...
-
Show the most stable conjugate base of these compounds: a) CHCHOH i b) HOCCHCHOH c) HNCHCHOH
-
Calculate the pKa for these compounds. a) HCOH (K 1.75 X 104) b) CHCH3 (K = 10-50)
-
In what phase must the moon be at the time of a solar eclipse? At the time of a lunar eclipse?
-
What are the gauge pressures at point A, B and C in each figure below? Water density is 1000 kg/m. The pressure po is the absolute pressure. Ambient pressure 101 325 Pa. Round off the answer to an...
-
1. The magnetic field now depends on location, according to the equation: B = Bo B = az a. Consider a thin strip of thickness dy that is at location zy. For this strip, write down expressions for B,...
-
The vector shown in Cartesian form is (-2687 +344 436 lbf O True False Y 3447) lbf 38 -X
-
Consider the falling cylinder viscometer as shown. Fluid of density, p whose viscosity is to be measured is placed in the tube, which stands vertically. The cylindrical block of density ps radius R...
-
Question 5 Composite Bzier surface Given the following control points of a bi-quadratic Bzier surface patch patch 1: 21 5); (18 23 0); (18 17 3); 17 6); (0 14 0); (8 14 6); (18 14 4); (0 (0 20 0); (8...
-
You might have heard of the run on Silicon Valley Bank (SVB) in the spring of 2023. This question explores that event. Banks have specialized balance sheets. Checking accounts and other deposits by...
-
Before the latest financial crisis and recession, when was the largest recession of the past 50 years, and what was the cumulative loss in output over the course of the slowdown?
-
Solve the inequality. |x| 3
-
Draw structures corresponding to the following names: (a) Bromoacetone (b) (S)-2-Hydroxypropanal (c) 2-Methyl-3-hcptanone (d) (2S, 3R)-2, 3, 4-Trihydroxybut anal (e) 2, 2, 4,...
-
Draw and name the seven aldehydes and ketones with the formula C5H10O. Which are chiral?
-
Give IUPAC names (or the following structures: (a) (b) (c) -- CH- "H (d) (e) (f) CH-CH CHH2H " C O:
-
- A two-dimensional vector makes an angle of 0 = 68.3 and has an x-component of 39.1 meters. Calculate the magnitude of this two- dimensional vector. Please enter a numerical answer below. Accepted...
-
a-1.If the required return is 11 percent, what is the profitability index for both projects? (Do not round intermediate calculations and round your answers to 3 decimal places, e.g., 32.161.) Project...
-
Perform the indicated operation (6x313x24x+5)=(x+1)

Study smarter with the SolutionInn App


