An Assign E or Z configuration to each of the following alkenes: ( - C (a) H2
Question:
An Assign E or Z configuration to each of the following alkenes:
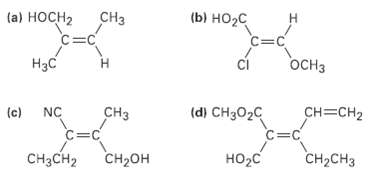
Transcribed Image Text:
(ы но-с CНз (a) НосH2 C=C C=C H3C ci OCH3 (c) (d) CH3O2C CH=CH2 NC CHз C=C CH3CH2 CH2он HO2C CH-CHз
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (14 reviews)
c High HOCH CH3 High CC Low HC ...View the full answer

Answered By

Ehsan Mahmood
I’ve earned Masters Degree in Business Studies and specialized in Accounts & Finance. Couple with this, I have earned BS Sociology from renowned institute of Pakistan. Moreover, I have humongous teaching experience at Graduate and Post-graduate level to Business and humanities students along with more than 7 years of teaching experience to my foreign students Online. I’m also professional writer and write for numerous academic journals pertaining to educational institutes periodically.
4.90+
248+ Reviews
287+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Assign a systematic (IUPAC) name to each of the following compounds: (a) (b) (c) (d) (e) Br Br
-
Assign a priority order to each of the following sets of groups: a. -CH(CH3)2, -CH3, -H, -NH2 b. -OH, -Br, -CH3, -CH2OH c. -OCH3, -NH(CH3)2, -CH2NH2, -OH d. -CH2CH2CH3, -CH2CH3, -C(CH3)3, -CH(CH3)2
-
Determine the configuration of each of the following alkenes as Z or E as appropriate: (a) (b) (c) H3C CH2CH2F CH2CH2OH (CH3)3 C -C CH3CH2 CH3
-
Calculate the heat required to raise 50 kg of solid sodium carbonate (Na2CO3) from 10C to 50C at 1atm using (a) The true heat capacity of Na2CO3, which is 1.14 kJ / (kg C). (b) A heat capacity...
-
What are your best-developed leadership traits, motives, and characteristics? How do you know?
-
Write the updating function associated with each of the following discrete-time dynamical systems and evaluate it at the given arguments. Which are linear? Qt+1 = 1/Qt + 1, evaluate at Qt = 0, Qt =...
-
Differentiate the cost and value drivers in a projects supply chain.
-
Romine Company issued $350,000 of 8%, 20-year bonds on January 1, 2014, at face value. Interest is payable annually on January 1. Instructions Prepare the journal entries to record the following...
-
Comparing Net Present Value with Internal Rate of Return 1. Starting from the Excel spreadsheet and using the =IRR(values,[guess]) Excel function, calculate the Internal Rate of Return (IRR) for...
-
Mountain Adventure Biking Park was started on May 1 by Dustin Tanner. The following events and transactions are for May: May 1 Tanner invested $70,000 cash in the business. 3 Purchased an out-of-use...
-
Rank the following sets of substituents in order of priority according to the Cahn-lngold?Prelog sequence rules: (a) -CH, -Br, -,-1 (b) -, -, -, -2 (c) -, -2CH3, -, - (d) -CH, -H2H, -H2H2, -H (e)...
-
Name the following cycloalkenes: (c) (b) (a) CH (f) (e) (d)
-
In problems 1-4, use Figure 2 and 3 approximate the indicated expressions. Figure 2 Figure 3 1. (f + g)' (4) 2. (f - 2g)'(2) 3. (fg)'(2) 4. (f/g)'(2) 111
-
Superior Limited's total overhead costs at various levels of activity are presented below: Month March April May June Machine Hours Total Overhead Costs 60,000 $216,800 50,000 194,000 70,000 239,600...
-
The density of a fluid changes with depth from the surface h according to the following formula: (h) = ch 2 , where c is a constant. A. What an expression for the gauge pressure p as a function of...
-
Helen holds 2,400 shares of Fizbo Incorporated stock that she purchased 11 months ago. The stock has done very well and has appreciated $22/share since Helen bought the stock. When sold, the stock...
-
A set S R^n is convex if for all x, y S and t [0, 1], we have (1 t)x + t y S (in other words, if S contains the points x and y, it contains the entire line segment between them). (a) If W R^n is a...
-
February 10 Activities Beginning inventory Purchase March 13 Purchase March 15 Sales August 21 Purchase September 5 Purchase September 10 Sales Totals Units Acquired at Cost 600 units @ $45 per unit...
-
What is ozone? How might Earth be affected if there were no ozone layer in the stratosphere?
-
Define the term utility software and give two examples.
-
Determine the kinds of intermolecular forces that are present in each element or compound. a. PH 3 b. HBr c. CH 3 OH d. I 2
-
In the addition of HBr to 3,3-dimethyl-l-butene, the results observed are shown in Fig. P5.36. (a) Explain why the different conditions give different product distributions. (b) Write a detailed...
-
Give the structures of both the reactive intermediate and the product in each of the following reactions: (a) (b) CH,CH,CCH, + HBr- CH2 CH,CH CCHH rides
-
The bromine atom in the bromonium ion in Eq. 5.13 (p. lg3) has a positive formal charge. Why can't bromins- utrdergo a Lewis acid=base association reaction with a nucleophito?
-
If employee costs were distributed equally for both WebLink and KH Connect, for which company, and for which year, was the highest employee cost per city?
-
S dx Evaluate ( +1) x(x+1)
-
Morganton Company makes one product and it provided the following information to help prepare the master budget: a. The budgeted selling price per unit is $65. Budgeted unit sales for June, July,...

Study smarter with the SolutionInn App


