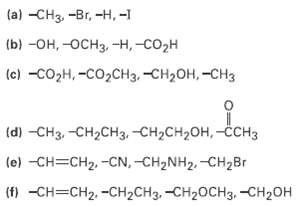
Rank the following sets of substituents in order of priority according to the Cahn-lngold?Prelog sequence rules: (a)
Question:
Rank the following sets of substituents in order of priority according to the Cahn-lngold?Prelog sequence rules:
Transcribed Image Text:
(a) -CHз, -Br, -Н,-1 (b) -он, -осНз, -Н, -СО2Н (c) -созн, -СО2CH3, -снОн, -снз (d) -CHз, -сH2сHз, -СH2сH2он, -ссHз (e) -CH3CH2 -CN, -CH2NH2. -Cн2Br () -сH-CH2. -СH2CH3. -СH20CH3. -СH2ОН
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
Highest priority a I Br CH3 H ...View the full answer

Answered By

Muhammad Haroon
More than 3 years experience in teaching undergraduate and graduate level courses which includes Object Oriented Programming, Data Structures, Algorithms, Database Systems, Theory of Automata, Theory of Computation, Database Administration, Web Technologies etc.
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Rank the following sets of substituents in order of Cahn-In-gold Prelog priorities: (a) CH3, 0H, H, C1 (b) CH3, CH2CH3, CH = CH2, CH2OH (c) CO2H, CH2OH, C = N, CH2NH2 (d) CH2CH3, C = CH, C = N,...
-
Order each of the following sets of compounds with respect to SN1reactivity: H3C CH3 C NH2 (a) i CH3CH2CHCH3 CI CH (b) (CH)3CI (CH)Br (CH)3C (c) Br CHCH3 C-Br CBr 3
-
Order each of the following sets of compounds with respect to SN2reactivity: CI CH (a) CH3CH2CHCH3 H CH3CH2CH2CI CH (b) CH CH CH CHCCH2Br CHH CH2Br Br CH (c) CH3CH2CH20CH3 CHCH2CH2Br CHCH2CH20Tos
-
The graph of f is given. (a) Why is f one-to-one? f is one-to-one because it passes the ---Select--- ? (b) What are the domain and range of f ? ?1 ?(Enter your answers in interval notation.) domain ?...
-
Provide an example of a leader you have appears to have good observed who cognitive intelligence, yet who is lacking in practical intelligence.
-
Fill in the missing percent, decimal, and/or fraction for each of the following: (Be careful! The total number of squares is now 801) EX Number of Squares Out of 80 75 Fraction Decimal Percent 15/16...
-
Describe the approaches to enhance value in the key phases of a projects supply chain.
-
Capital Lease. The City of Jamestown has agreed to acquire a new city maintenance building under a capital lease agreement. At the inception of the lease, a payment of $100,000 is to be made: nine...
-
Hedging EUR currency risk from the US perspective Terabit Inc is an American exporting company of electronic components to countries in the Euro area. The company mainly sells its products in Euro,...
-
Ceramic Structures has experienced rapid growth over the past several years. Sales are expected to grow at 15% per year for the next three years. Sales growth has been fueled by aggressive pricing as...
-
Predict the major product from addition of HBr to each of the following alkenes: (a) CH2 (b) (c) CH CH3CHHHH
-
An Assign E or Z configuration to each of the following alkenes: ( - C (a) H2 C=C C=C H3C ci OCH3 (c) (d) CH3O2C CH=CH2 NC CH C=C CH3CH2 CH2 HO2C CH-CH
-
You have won a free trip on the Queen Elizabeth II and are in mid-Atlantic steaming due east at 45 km/h as the Concorde passes directly overhead flying due west at Mach 1.6 at an altitude of 12,500...
-
(2). A minimization LP is being solved by the big M method. e is the excess variable in constraint 1, s2 is the slack variable in constraint 2, and a, a3 are the artificial variables of constraints...
-
315 g of a solution has a volume of 4.2 L. 5 L of this solution sells for $9.29. At these rates, what mass of the solution can we buy with $120? a. (1/10 marks) List the unknown quantity name,...
-
7. Solve and give physical interpretations of the problems Vy(x,y)=0, 0
-
You pay $1,000 to flip a two-sided, fair coin at the local fair. If you flip 'heads', you walk away with $3,000, a return of 200%. However, if you flip 'tails', you walk away with $250, a return of...
-
A researcher believes that 48% of people who grew up as the only child have an IQ score over 100. However, unknown to the researcher, this figure is actually 50% , which is the same as in the general...
-
What role does the Sun play in ocean currents?
-
A report from the college dean indicates that for the previous semester, the grade distribution for the Department of Psychology included 135 As, 158 Bs, 140 Cs, 94 Ds, and 53 Fs. Determine what kind...
-
Arrange these compounds in order of increasing boiling point. Explain your reasoning. a. H 2 S b. H 2 Se c. H 2 O
-
Deuterium (D, or 2H; is an isotope of hydrogen with atomic mass - 2. Deuterium can be introduced into organic compounds by using reagents in which hydrogen has been replaced by deuterium. Outline...
-
Deuterium (D, or 2H; is an isotope of hydrogen with atomic mass - 2. Deuterium can be introduced into organic compounds by using reagents in which hydrogen has been replaced by deuterium. Outline...
-
In the addition of HBr to 3,3-dimethyl-l-butene, the results observed are shown in Fig. P5.36. (a) Explain why the different conditions give different product distributions. (b) Write a detailed...
-
Research: What is a sovereign nation, how many sovereign nations are recognized as members by the United Nations and how many sovereign nations are recognized by the US State Department. Compare the...
-
Marigold Corp. purchases a patent for $198,000 on January 2, 2022. Its estimated useful life is 18 years. (a) Prepare the journal entry to record amortization expense for the first year. (Credit...
-
With an interest of $ 2 4 . 3 1 and a principal of $ 3 , 5 0 0 for 1 0 0 days, using the ordinary interest method, the rate is ?

Study smarter with the SolutionInn App


